Estrogen Receptor and Vascular Aging
- PMID: 35821994
- PMCID: PMC9261451
- DOI: 10.3389/fragi.2021.727380
Estrogen Receptor and Vascular Aging
Abstract
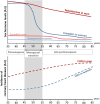
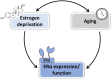
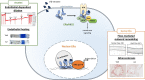
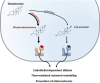
Cardiovascular diseases remain an age-related pathology in both men and women. These pathologies are 3-fold more frequent in men than in women before menopause, although this difference progressively decreases after menopause. The vasculoprotective role of estrogens are well established before menopause, but the consequences of their abrupt decline on the cardiovascular risk at menopause remain debated. In this review, we will attempt to summarize the main clinical and experimental studies reporting the protective effects of estrogens against cardiovascular diseases, with a particular focus on atherosclerosis, and the impact of aging and estrogen deprivation on their endothelial actions. The arterial actions of estrogens, but also part of that of androgens through their aromatization into estrogens, are mediated by the estrogen receptor (ER)α and ERβ. ERs belong to the nuclear receptor family and act by transcriptional regulation in the nucleus, but also exert non-genomic/extranuclear actions. Beside the decline of estrogens at menopause, abnormalities in the expression and/or function of ERs in the tissues, and particularly in arteries, could contribute to the failure of classic estrogens to protect arteries during aging. Finally, we will discuss how recent insights in the mechanisms of action of ERα could contribute to optimize the hormonal treatment of the menopause.
Keywords: atherosclerosis; endothelium; estradiol; estrogen receptor; menopause; vascular aging.
Copyright © 2021 Davezac, Buscato, Zahreddine, Lacolley, Henrion, Lenfant, Arnal and Fontaine.
Conflict of interest statement
The laboratory received support from Mithra Pharmaceuticals for preclinical studies on estetrol.
Figures
References
-
- Abot A., Fontaine C., Buscato M., Solinhac R., Flouriot G., Fabre A., et al. (2014). The Uterine and Vascular Actions of Estetrol Delineate a Distinctive Profile of Estrogen Receptor α Modulation, Uncoupling Nuclear and Membrane Activation. EMBO Mol. Med. 6, 1328–1346. 10.15252/emmm.201404112 - DOI - PMC - PubMed
-
- Adlanmerini M., Fébrissy C., Zahreddine R., Vessières E., Buscato M., Solinhac R., et al. (2020). Mutation of Arginine 264 on ERα (Estrogen Receptor Alpha) Selectively Abrogates the Rapid Signaling of Estradiol in the Endothelium without Altering Fertility. Atvb 40, 2143–2158. 10.1161/ATVBAHA.120.314159 - DOI - PubMed
-
- Adlanmerini M., Solinhac R., Abot A., Fabre A., Raymond-Letron I., Guihot A.-L., et al. (2014). Mutation of the Palmitoylation Site of Estrogen Receptor In Vivo Reveals Tissue-specific Roles for Membrane versus Nuclear Actions. Proc. Natl. Acad. Sci. 111, E283–E290. 10.1073/pnas.1322057111 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

