Microvascular Sex- and Age- Dependent Phosphodiesterase Expression
- PMID: 35822023
- PMCID: PMC9261398
- DOI: 10.3389/fragi.2021.719698
Microvascular Sex- and Age- Dependent Phosphodiesterase Expression
Abstract
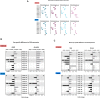
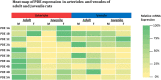
Objective: The cyclic nucleotide second messengers, cAMP and cGMP, are pivotal regulators of vascular functions; their cellular levels are tightly controlled by the cyclic nucleotide hydrolases, phosphodiesterases (PDE). Biologic sex and age are recognized as independent factors impacting the mechanisms mediating both vascular health and dysfunction. This study focused on microvessels isolated from male and female rats before (juvenile) and after (adult) sexual maturity under resting conditions. We tested the hypothesis that sexual dimorphism in microvascular PDE expression would be absent in juvenile rats, but would manifest in adult rats. Methods: Abdominal skeletal muscle arterioles and venules were isolated from age-matched juvenile and adult male and female rats under resting conditions. Transcripts of five PDE families (1-5) associated with coronary and vascular function with a total of ten genes were measured using TaqMan real-time RT-PCR and protein expression of microvessel PDE4 was assessed using immunoblotting and immunofluorescence. Results: Overall expression levels of PDE5A were highest while PDE3 levels were lowest among the five PDE families (p < 0.05) regardless of age or sex. Contrary to our hypothesis, in juveniles, sexual dimorphism in PDE expression was observed in three genes: arterioles (PDE1A, female > male) and venules (PDE1B and 3A, male > female). In adults, gene expression levels in males were higher than females for five genes in arterioles (PDE1C, 3A, 3B, 4B, 5A) and three genes (PDE3A, 3B, and 5A) in venules. Furthermore, age-related differences were observed in PDE1-5 (in males, adult > juvenile for most genes in arterioles; in females, adult > juvenile for arteriolar PDE3A; juvenile gene expression > adult for two genes in arterioles and three genes in venules). Immunoblotting and immunofluorescence analysis revealed protein expression of microvessel PDE4. Conclusion: This study revealed sexual dimorphism in both juvenile and adult rats, which is inconsistent with our hypothesis. The sex- and age-dependent differences in PDE expression implicate different modulations of cAMP and cGMP pathways for microvessels in health. The implication of these sex- and age-dependent differences, as well as the duration and microdomain of PDE1-5 activities in skeletal muscle microvessels, in both health and disease, require further investigation.
Keywords: adult; age; arterioles; juvenile; phosphodiesterase; sex; skeletal muscle; venules.
Copyright © 2021 Wang, Kazmi and Huxley.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Intrinsic sex-specific differences in microvascular endothelial cell phosphodiesterases.Am J Physiol Heart Circ Physiol. 2010 Apr;298(4):H1146-54. doi: 10.1152/ajpheart.00252.2009. Epub 2010 Feb 5. Am J Physiol Heart Circ Physiol. 2010. PMID: 20139324 Free PMC article.
-
PDE4 and PDE5 regulate cyclic nucleotides relaxing effects in human umbilical arteries.Eur J Pharmacol. 2008 Mar 17;582(1-3):102-9. doi: 10.1016/j.ejphar.2007.12.017. Epub 2007 Dec 28. Eur J Pharmacol. 2008. PMID: 18234184
-
Presence of cyclic nucleotide phosphodiesterases PDE1A, existing as a stable complex with calmodulin, and PDE3A in human spermatozoa.Biol Reprod. 2002 Aug;67(2):423-30. doi: 10.1095/biolreprod67.2.423. Biol Reprod. 2002. PMID: 12135876
-
Cyclic nucleotide phosphodiesterases in heart and vessels: A therapeutic perspective.Arch Cardiovasc Dis. 2016 Jun-Jul;109(6-7):431-43. doi: 10.1016/j.acvd.2016.02.004. Epub 2016 May 13. Arch Cardiovasc Dis. 2016. PMID: 27184830 Review.
-
Phosphodiesterase 1: A Unique Drug Target for Degenerative Diseases and Cognitive Dysfunction.Adv Neurobiol. 2017;17:349-384. doi: 10.1007/978-3-319-58811-7_13. Adv Neurobiol. 2017. PMID: 28956339 Review.
Cited by
-
Dysregulated Cyclic Nucleotide Metabolism in Alcohol-Associated Steatohepatitis: Implications for Novel Targeted Therapies.Biology (Basel). 2023 Oct 10;12(10):1321. doi: 10.3390/biology12101321. Biology (Basel). 2023. PMID: 37887031 Free PMC article.
-
Sex-Specific Susceptibility Loci Associated With Coronary Artery Aneurysms in Patients With Kawasaki Disease.Korean Circ J. 2024 Sep;54(9):577-586. doi: 10.4070/kcj.2023.0244. Epub 2024 Apr 8. Korean Circ J. 2024. PMID: 38767439 Free PMC article.
References
-
- Applied Biosystem (2008). Guide to Performing Relative Quantitation of Gene Expression Using Real-Time Quantitative PCR, 56.
-
- Cai Y., Nagel D. J., Zhou Q., Cygnar K. D., Zhao H., Li F., et al. (2015). Role of cAMP-Phosphodiesterase 1C Signaling in Regulating Growth Factor Receptor Stability, Vascular Smooth Muscle Cell Growth, Migration, and Neointimal Hyperplasia. Circ. Res. 116 (7), 1120–1132. 10.1161/CIRCRESAHA.116.304408 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

