Obesity Accelerates Age-Associated Defects in Human B Cells Through a Metabolic Reprogramming Induced by the Fatty Acid Palmitate
- PMID: 35822047
- PMCID: PMC9261304
- DOI: 10.3389/fragi.2021.828697
Obesity Accelerates Age-Associated Defects in Human B Cells Through a Metabolic Reprogramming Induced by the Fatty Acid Palmitate
Abstract
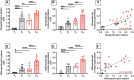
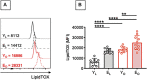
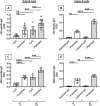
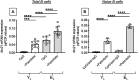
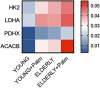
We have measured the secretion of autoimmune antibodies in plasma samples and in culture supernatants of blood-derived B cells from four groups of individuals: young lean (YL), elderly lean (EL), young obese (YO) and elderly obese (EO). We found secretion comparable in YO and EL individuals, suggesting that obesity accelerates age-associated defects in circulating B cells. To define at least one possible molecular pathway involved, we used an in vitro model in which B cells from YL and EL individuals have been stimulated with the Fatty Acid (FA) palmitate, the most common saturated FA in the human body. The rationale to use palmitate is that there is a chronic increase in circulating levels of palmitate, due to increased spontaneous lipolysis occurring during aging and obesity, and this may induce autoimmune B cells. Results herein show that in vitro incubation of B cells from YL and EL individuals with the FA palmitate induces mRNA expression of T-bet, the transcription factor for autoimmune antibodies, as well as secretion of autoimmune IgG antibodies, with B cells from YL individuals looking similar to B cells from EL individuals, confirming our initial hypothesis. The generation of autoimmune B cells in the presence of the FA palmitate was found to be associated with a metabolic reprogramming of B cells from both YL and EL individuals. These results altogether show the critical role of the FA palmitate in inducing human B cell immunosenescence and show for the first time the importance of metabolic pathways in this process.
Keywords: B cells; aging; autoimmunity; metabolism; obesity.
Copyright © 2022 Frasca, Romero, Garcia, Diaz and Blomberg.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
LinkOut - more resources
Full Text Sources

