Follicular Hyperstimulation Dysgenesis: New Explanation for Adverse Effects of Excessive FSH in Ovarian Stimulation
- PMID: 35833461
- PMCID: PMC9342683
- DOI: 10.1210/endocr/bqac100
Follicular Hyperstimulation Dysgenesis: New Explanation for Adverse Effects of Excessive FSH in Ovarian Stimulation
Abstract
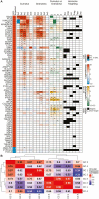
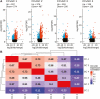
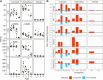
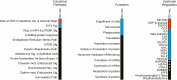
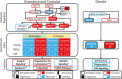
High follicle-stimulating hormone (FSH) doses during ovarian stimulation protocols for assisted reproductive technologies (ART) are detrimental to ovulatory follicle function and oocyte quality. However, the mechanisms are unclear. In a small ovarian reserve heifer model, excessive FSH doses lead to phenotypic heterogeneity of ovulatory size follicles, with most follicles displaying signs of premature luteinization and a range in severity of abnormalities. By performing whole transcriptome analyses of granulosa cells, cumulus cells, and oocytes from individual follicles of animals given standard or excessive FSH doses, we identified progressive changes in the transcriptomes of the 3 cell types, with increasing severity of follicular abnormality with the excessive doses. The granulosa and cumulus cells each diverged progressively from their normal phenotypes and became highly similar to each other in the more severely affected follicles. Pathway analysis indicates a possible dysregulation of the final stages of folliculogenesis, with processes characteristic of ovulation and luteinization occurring concurrently rather than sequentially in the most severely affected follicles. These changes were associated with disruptions in key pathways in granulosa and cumulus cells, which may account for previously reported reduced estradiol production, enhanced progesterone and oxytocin production and diminished ovulation rates. Predicted deficiencies in oocyte survival, stress response, and fertilization suggest likely reductions in oocyte health, which could further compromise oocyte quality and ART outcomes.
Keywords: cumulus cells; excessive FSH doses; granulosa cells; oocytes; ovarian stimulation; ovulatory-size follicles; transcriptome analysis.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Endocrine Society. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures


References
-
- Clark ZL, Thakur M, Leach RE, Ireland JJ. FSH dose is negatively correlated with number of oocytes retrieved: analysis of a data set with ~650,000 ART cycles that previously identified an inverse relationship between FSH dose and live birth rate. J Assist Reprod and Genet. 2021;38(7):1787-1797. - PMC - PubMed
-
- McGowan MR, Braithwaite M, Jochle W, Mapletoft RJ. Superovulation of beef heifers with Pergonal (HMG): a dose response trial. Theriogenology. 1985;24(2):173-184. - PubMed
-
- Pawlyshyn V, Lindsell CE, Braithwaite M, Mapletoft RJ. Superovulation of beef cows with FSH-P: a dose-response trial. Theriogenology. 1986;25(1):179.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

