Biodegradable magnesium alloys for orthopaedic applications
- PMID: 35836650
- PMCID: PMC9255811
- DOI: 10.12336/biomatertransl.2021.03.005
Biodegradable magnesium alloys for orthopaedic applications
Abstract
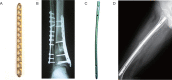
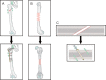
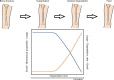
There is increasing interest in the development of bone repair materials for biomedical applications. Magnesium (Mg)-based alloys have a natural ability to biodegrade because they corrode in aqueous media; they are thus promising materials for orthopaedic device applications in that the need for a secondary surgical operation to remove the implant can be eliminated. Notably, Mg has superior biocompatibility because Mg is found in the human body in abundance. Moreover, Mg alloys have a low elastic modulus, close to that of natural bone, which limits stress shielding. However, there are still some challenges for Mg-based fracture fixation. The degradation of Mg alloys in biological fluids can be too rapid, resulting in a loss of mechanical integrity before complete healing of the bone fracture. In order to achieve an appropriate combination of bio-corrosion and mechanical performance, the microstructure needs to be tailored properly by appropriate alloy design, as well as the use of strengthening processes and manufacturing techniques. This review covers the evolution, current strategies and future perspectives of Mg-based orthopaedic implants.
Keywords: biodegradability; magnesium alloys; mechanical behaviour; microstructure; orthopaedic application.
Figures



References
-
- Ratner B. D., Hoffman A. S., Schoen F. J., Lemons J. E. Biomaterial sciences: an introduction to materials in medicine. Academic Press; San Diego: 1996.
-
- Temenoff J. S., Mikos A. G. Biomaterials: the intersection of biology and materials sceince. Pearson; 2008.
-
- Zheng Y. F., Gu X. N., Witte F. Biodegradable metals. Mater Sci Eng R Rep. 2014;77:1–34.
-
- Ermanno B. Basic Composition and Structure of Bone. In: An Y. H., Draughn R. A., editors. Mechanical testing of bone and the bone-implant interface. CRC Press; Boca Raton: 1999. pp. 3–22.
-
- Suzuki H., He J. IOP Conference Series: Materials Science and Engineering. Vol. 269. IOP Publishing; Tianjin: 2017. Evaluation on bending properties of biomaterial GUM Metal meshed plates for bone graft applications; 012078 pp.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
