Metronidazole-loaded nanoparticulate thermoreversible gel for gynecologic infection of Trichomonas vaginalis
- PMID: 35836901
- PMCID: PMC9274572
Metronidazole-loaded nanoparticulate thermoreversible gel for gynecologic infection of Trichomonas vaginalis
Abstract
Objective: Trichomoniasis is a common sexually-transmitted disease that is associated with increased perinatal morbidity and human immunodeficiency virus (HIV) transmission. This study aimed to develop a Metronidazole-loaded nanoparticulate thermoreversible gel for gynecological infection of Trichomonas vaginalis (T. vaginalis).
Methods: The optimized nanoparticulate formulation was used in thermoreversible gel and characterized for physico-chemical properties, antiparasitic activity, and in vivo efficacy in the BALB/c mouse model.
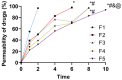
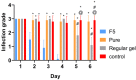
Result: A nearly threefold rise in antiparasitic activity of the optimized formulation was observed as compared to that of regular gel. Formulation F5 successfully cured the trichomoniasis within 3 days, while regular gel and pure Metronidazole (MTDZ) failed to cure this infection (P<0.05).
Conclusion: The present investigation confirms the ability of thermoreversible gel containing nanoparticulate metronidazole againstthe infection by T. vaginalis. The developed gel could be an alternative to the existing drug delivery system for the treatment of trichomoniasis.
Keywords: Metronidazole; Trichomonas vaginalis; chitosan; drug delivery; nanoparticles; poloxamer; thermoreversible; trichomoniasis; vaginal.
AJTR Copyright © 2022.
Conflict of interest statement
None.
Figures

References
-
- Fernando HV, Chan LL, Dang N, Santhanes D, Banneheke H, Nalliah S, Coombes AGA. Controlled delivery of the antiprotozoal agent (tinidazole) from intravaginal polymer matrices for treatment of the sexually transmitted infection, trichomoniasis. Pharm Dev Technol. 2019;24:348–356. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
