Probing Neuro-Endocrine Interactions Through Remote Magnetothermal Adrenal Stimulation
- PMID: 35837128
- PMCID: PMC9274974
- DOI: 10.3389/fnins.2022.901108
Probing Neuro-Endocrine Interactions Through Remote Magnetothermal Adrenal Stimulation
Abstract
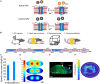
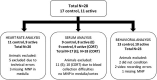
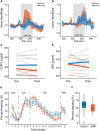
Exposure to stressful or traumatic stimuli may alter hypothalamic-pituitary-adrenal (HPA) axis and sympathoadrenal-medullary (SAM) reactivity. This altered reactivity may be a component or cause of mental illnesses. Dissecting these mechanisms requires tools to reliably probe HPA and SAM function, particularly the adrenal component, with temporal precision. We previously demonstrated magnetic nanoparticle (MNP) technology to remotely trigger adrenal hormone release by activating thermally sensitive ion channels. Here, we applied adrenal magnetothermal stimulation to probe stress-induced HPA axis and SAM changes. MNP and control nanoparticles were injected into the adrenal glands of outbred rats subjected to a tone-shock conditioning/extinction/recall paradigm. We measured MNP-triggered adrenal release before and after conditioning through physiologic (heart rate) and serum (epinephrine, corticosterone) markers. Aversive conditioning altered adrenal function, reducing corticosterone and blunting heart rate increases post-conditioning. MNP-based organ stimulation provides a novel approach to probing the function of SAM, HPA, and other neuro-endocrine axes and could help elucidate changes across stress and disease models.
Keywords: adrenal gland; corticosterone; epinephrine; hormones; magnetic nanoparticles; neuromodulation.
Copyright © 2022 Maeng, Rosenfeld, Simandl, Koehler, Senko, Moon, Varnavides, Murillo, Reimer, Wald, Anikeeva and Widge.
Conflict of interest statement
ASW, PA, and DR are inventors on a patent application for therapeutic uses of controlled adrenal release. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Early life stress in rats sex-dependently affects remote endocrine rather than behavioral consequences of adult exposure to contextual fear conditioning.Horm Behav. 2018 Jul;103:7-18. doi: 10.1016/j.yhbeh.2018.05.017. Epub 2018 May 30. Horm Behav. 2018. PMID: 29802874
-
Adrenal axis activation by chronic social stress fails to inhibit gonadal function in male rats.Psychoneuroendocrinology. 1997 Nov;22(8):563-73. doi: 10.1016/s0306-4530(97)00051-6. Psychoneuroendocrinology. 1997. PMID: 9483702
-
Experimentally-induced hyperthyroidism is associated with activation of the rat hypothalamic-pituitary-adrenal axis.Eur J Endocrinol. 2005 Jul;153(1):177-85. doi: 10.1530/eje.1.01923. Eur J Endocrinol. 2005. PMID: 15994759
-
Hypothalamic-pituitary-adrenal and hypothalamic-pituitary-gonadal axes: sex differences in regulation of stress responsivity.Stress. 2017 Sep;20(5):476-494. doi: 10.1080/10253890.2017.1369523. Epub 2017 Aug 31. Stress. 2017. PMID: 28859530 Free PMC article. Review.
-
Activation of the hypothalamic-pituitary-adrenal axis in lithium-induced conditioned taste aversion learning.Eur J Pharmacol. 2015 Dec 5;768:182-8. doi: 10.1016/j.ejphar.2015.10.052. Epub 2015 Oct 30. Eur J Pharmacol. 2015. PMID: 26524411 Review.
Cited by
-
Modulating cell signalling in vivo with magnetic nanotransducers.Nat Rev Methods Primers. 2022;2:92. doi: 10.1038/s43586-022-00170-2. Epub 2022 Nov 17. Nat Rev Methods Primers. 2022. PMID: 38111858 Free PMC article.
References
-
- American Psychiatric Association [APA] (2013). Diagnostic And Statistical Manual of Mental Disorders, 5th Edn. Washington, DC: American Psychiatric Association.
-
- Bystritsky A., Korb A. S. (2015). A Review of Low-Intensity Transcranial Focused Ultrasound for Clinical Applications. Curr. Behav. Neurosci. Rep. 2 60–66. 10.1007/s40473-015-0039-0 - DOI
LinkOut - more resources
Full Text Sources

