Diverse actions of sirtuin-1 on ovulatory genes and cell death pathways in human granulosa cells
- PMID: 35840944
- PMCID: PMC9284863
- DOI: 10.1186/s12958-022-00970-x
Diverse actions of sirtuin-1 on ovulatory genes and cell death pathways in human granulosa cells
Abstract
Background: Human granulosa-lutein cells (hGLCs) amply express sirtuin-1 (SIRT1), a NAD + -dependent deacetylase that is associated with various cellular functions. SIRT1 was shown to elevate cAMP on its own and additively with human chorionic gonadotropin (hCG), it is therefore interesting to examine if SIRT1 affects other essential hGLC functions.
Methods: Primary hGLCs, obtained from the follicular aspirates of women undergoing IVF and SV40-transfected, immortalized hGLCs (SVOG cells), were used. Primary cells were treated with SIRT1 specific activator SRT2104, as well as hCG or their combination. Additionally, siRNA-targeting SIRT1 construct was used to silence endogenous SIRT1 in SVOG cells. PTGS2, EREG, VEGFA and FGF2 expression was determined using quantitative polymerase chain reaction (qPCR). Apoptotic and necroptotic proteins were determined by specific antibodies in western blotting. Cell viability/apoptosis was determined by the XTT and flow cytometry analyses. Data were analyzed using student t-test or Mann-Whitney U test or one-way ANOVA followed by Tukey HSD post hoc test.
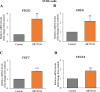
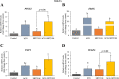
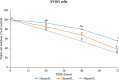
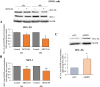
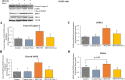
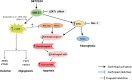
Results: In primary and immortalized hGLCs, SRT2104 significantly upregulated key ovulatory and angiogenic genes: PTGS2, EREG, FGF2 and VEGFA, these effects tended to be further augmented in the presence of hCG. Additionally, SRT2104 dose and time-dependently decreased viable cell numbers. Flow cytometry of Annexin V stained cells confirmed that SIRT1 reduced live cell numbers and increased late apoptotic and necrotic cells. Moreover, we found that SIRT1 markedly reduced anti-apoptotic BCL-XL and MCL1 protein levels and increased cleaved forms of pro-apoptotic proteins caspase-3 and PARP. SIRT1 also significantly induced necroptotic proteins RIPK1 and MLKL. RIPK1 inhibitor, necrostatin-1 mitigated SIRT1 actions on RIPK1 and MLKL but also on cleaved caspase-3 and PARP and in accordance on live and apoptotic cells, implying a role for RIPK1 in SIRT1-induced cell death. SIRT1 silencing produced inverse effects on sorted cell populations, anti-apoptotic, pro-apoptotic and necroptotic proteins, corroborating SIRT1 activation.
Conclusions: These findings reveal that in hGLCs, SIRT1 enhances the expression of ovulatory and angiogenic genes while eventually advancing cell death pathways. Interestingly, these seemingly contradictory events may have occurred in a cAMP-dependent manner.
Keywords: Angiogenesis; Apoptosis; Necroptosis; Ovulation.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

