Changes in somatosensory evoked potentials elicited by lateral cerebellar nucleus deep brain stimulation in the naïve rodent
- PMID: 35842210
- PMCID: PMC10966384
- DOI: 10.1016/j.neulet.2022.136800
Changes in somatosensory evoked potentials elicited by lateral cerebellar nucleus deep brain stimulation in the naïve rodent
Abstract
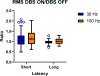
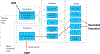
Deep brain stimulation (DBS) of the deep cerebellar nuclei has been shown to enhance perilesional cortical excitability and promote motor rehabilitation in preclinical models of cortical ischemia and is currently being evaluated in patients with chronic, post-stroke deficits. Understanding the effects of cerebellar DBS on contralateral sensorimotor cortex may be key to developing approaches to optimize stimulation delivery and treatment outcomes. Using the naïve rat model, we characterized the effects of DBS of the lateral cerebellar nucleus (LCN) on somatosensory evoked potentials (SSEPs) and evaluated their potential use as a surrogate index of cortical excitability. SSEPs were recorded concurrently with continuous 30 Hz or 100 Hz LCN DBS and compared to the DBS OFF condition. Ratios of SSEP peak to peak amplitude during 100 Hz LCN DBS to DBS OFF at longer latency peaks were significantly>1, suggesting that cortical excitability was enhanced as a result of LCN DBS. Although changes in SSEP peak to peak amplitudes were observed, they were modest in relation to previously reported effects on motor cortical excitability. Overall, our findings suggest that LCN output influences thalamocortical somatosensory pathways, however further work is need to better understand the potential role of SSEPs in optimizing therapy.
Keywords: Cortical excitability; Deep brain stimulation; Dentatothalamocortical pathway; Somatosensory evoked potentials; Stroke rehabilitation.
Copyright © 2022 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest:
Drs. Machado and Baker have intellectual property and distribution rights in Enspire DBS Therapy, Inc., which is a spin-off of the Cleveland Clinic. Drs. Machado and Baker serve on the Scientific Advisory Board of Enspire DBS Therapy, Inc. Dr. Machado holds a patent titled as “Methods of treating medical conditions by neuromodulation of the cerebellar pathways.” US 7,640,063.
Figures

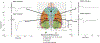


References
-
- Ward NS, Restoring brain function after stroke—bridging the gap between animals and humans, Nat Rev Neurol 13 (2017) 244–255. - PubMed
-
- Hummel F, Cohen LG, Improvement of motor function with noninvasive cortical stimulation in a patient with chronic stroke, Neurorehabilitation and neural repair 19 (2005) 14–19. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

