Hyperactivity of Purkinje cell and motor deficits in C9orf72 knockout mice
- PMID: 35843530
- PMCID: PMC10369482
- DOI: 10.1016/j.mcn.2022.103756
Hyperactivity of Purkinje cell and motor deficits in C9orf72 knockout mice
Abstract
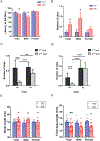
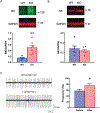
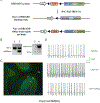
A hexanucleotide (GGGGCC) repeat expansion in the first intron of the C9ORF72 gene is the most frequently reported genetic cause of amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). The cerebellum has not traditionally been thought to be involved in the pathogenesis of C9ORF72-associated ALS/FTD, but recent evidence suggested a potential role. C9ORF72 is highly expressed in the cerebellum. Decreased C9ORF72 transcript and protein levels were detected in the postmortem cerebellum, suggesting a loss-of-function effect of C9ORF72 mutation. This study investigated the role of loss of C9ORF72 function using a C9orf72 knockout mouse line. C9orf72 deficiency led to motor impairment in rotarod, beam-walking, paw-print, open-field, and grip-strength tests. Purkinje cells are the sole output neurons in the cerebellum, and we next determined their involvement in the motor phenotypes. We found hyperactivity of Purkinje cells in the C9orf72 knockout mouse accompanied by a significant increase of the large-conductance calcium-activated potassium channel (BK) protein in the cerebellum. The link between BK and Purkinje cell firing was demonstrated by the acute application of the BK activator that increased the firing frequency of the Purkinje cells ex vivo. In vivo chemogenetic activation of Purkinje cells in wild-type mice led to similar motor deficits in rotarod and beam-walking tests. Our results highlight that C9ORF72 loss alters the activity of the Purkinje cell and potentially the pathogenesis of the disease. Manipulating the Purkinje cell firing or cerebellar output may contribute to C9ORF72-associated ALS/FTD treatment.
Keywords: BK channel; C9orf72; Hyperactivity; Motor behavior; Purkinje cell.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures
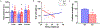

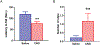
References
-
- Arai T, Hasegawa M, Akiyama H, Ikeda K, Nonaka T, Mori H, … Oda T (2006). TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun, 351(3), 602–611. 10.1016/j.bbrc.2006.10.093 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

