Population Pharmacokinetic Modeling of Lucitanib in Patients with Advanced Cancer
- PMID: 35844029
- PMCID: PMC9399017
- DOI: 10.1007/s13318-022-00773-w
Population Pharmacokinetic Modeling of Lucitanib in Patients with Advanced Cancer
Abstract
Background: Lucitanib is an oral, potent, selective inhibitor of the tyrosine kinase activity of vascular endothelial growth factor receptors 1‒3, fibroblast growth factor receptors 1‒3, and platelet-derived growth factor receptors alpha/beta.
Objective: We aimed to develop a population pharmacokinetics (PopPK) model for lucitanib in patients with advanced cancers.
Methods: PopPK analyses were based on intensive and sparse oral pharmacokinetic data from 5 phase 1/2 clinical studies of lucitanib in a total of 403 patients with advanced cancers. Lucitanib was administered at 5‒30 mg daily doses as 1 of 2 immediate-release oral formulations: a film-coated tablet or a hard gelatin capsule.
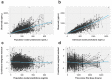
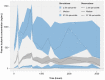
Results: Lucitanib pharmacokinetics were best described by a 2-compartment model with zero-order release into the dosing compartment, followed by first-order absorption and first-order elimination. Large between-subject pharmacokinetic variability was partially explained by body weight. No effects of demographics or tumor type on lucitanib pharmacokinetics were observed. The model suggested that the formulation impacted release duration (tablet, 0.243 h; capsule, 0.814 h), but the effect was not considered clinically meaningful. No statistically significant effects were detected for concomitant cytochrome P450 (CYP) 3A4 inhibitors or inducers, CYP2C8 or P-glycoprotein inhibitors, serum albumin, mild/moderate renal impairment, or mild hepatic impairment. Concomitant proton pump inhibitors had no clinically significant effect on lucitanib absorption.
Conclusions: The PopPK model adequately described lucitanib pharmacokinetics. High between-subject pharmacokinetic variability supports a safety-based dose-titration strategy currently being used in an ongoing clinical study of lucitanib to optimize drug exposure and clinical benefit.
Trial registration: ClinicalTrials.gov Identifier: NCT01283945, NCT02053636, ISRCTN23201971, NCT02202746, NCT02109016.
© 2022. The Author(s).
Conflict of interest statement
Mingxiang Liao, Kenton Wride, Denise Lepley, Terri Cameron, and Jim Xiao are or were employees of Clovis Oncology, Inc. Mark Sale and Jie Zhou are or were paid consultants for Clovis Oncology, Inc.
Figures



References
-
- Dusek RL, Robillard L, Harding TC, Simmons AD, Nguyen M. Abstract 1214: enhancement of anti-PD-1 antitumor efficacy in syngenic preclinical models by the angiogenesis inhibitor lucitanib. Cancer Res. 2019;79:1214. doi: 10.1158/1538-7445.AM2019-1214. - DOI
-
- Soria J, De Braud F, Cereda R, Bahleda R, Delmonte A, Angevin E, et al. First-in-man study of E-3810, a novel VEGFR and FGFR inhibitor, in patients with advanced solid tumors. J Clin Oncol. 2011;29:TPS149. doi: 10.1200/jco.2011.29.15_suppl.tps149. - DOI
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

