Two Main Cellular Components in Rheumatoid Arthritis: Communication Between T Cells and Fibroblast-Like Synoviocytes in the Joint Synovium
- PMID: 35844494
- PMCID: PMC9284267
- DOI: 10.3389/fimmu.2022.922111
Two Main Cellular Components in Rheumatoid Arthritis: Communication Between T Cells and Fibroblast-Like Synoviocytes in the Joint Synovium
Abstract
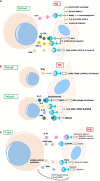
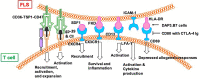
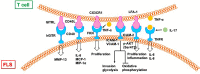
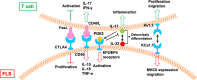
Rheumatoid arthritis (RA) is a chronic autoimmune disease that endangers the health of approximately 1% of the global population. Current RA medications on the market mainly include non-steroidal anti-inflammatory drugs, biological agents, and disease-modifying drugs. These drugs aim to inhibit the overactivated immune response or inflammation of RA, but they cannot cure RA. A better understanding of the pathogenesis of RA will provide a new understanding to search for RA targets and for drug development. The infiltration of T cells and hyper-proliferation of fibroblast-like synoviocytes (FLS) in the synovium of patients with RA are significantly upregulated. Furthermore, the abnormal activation of these two types of cells has been confirmed to promote development of the course of A by many studies. This article systematically summarizes the interactions between T cells and FLS in RA synovial tissues, including one-way/mutual regulation and direct/indirect regulation between the two. It further aims to investigate the pathogenesis of RA from the perspective of mutual regulation between T cells and FLS and to provide new insights into RA research.
Keywords: FLS; RA; T cells; cellular interaction; therapy.
Copyright © 2022 Tu, Huang, Zhang, Mei and Zhu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

