Multifaceted Roles of the E3 Ubiquitin Ligase RING Finger Protein 115 in Immunity and Diseases
- PMID: 35844553
- PMCID: PMC9279554
- DOI: 10.3389/fimmu.2022.936579
Multifaceted Roles of the E3 Ubiquitin Ligase RING Finger Protein 115 in Immunity and Diseases
Abstract
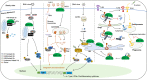
Ubiquitination is a post-translational modification that plays essential roles in various physiological and pathological processes. Protein ubiquitination depends on E3 ubiquitin ligases that catalyze the conjugation of ubiquitin molecules on lysine residues of targeted substrates. RING finger protein 115 (RNF115), also known as breast cancer associated gene 2 (BCA2) and Rab7-interacting RING finger protein (Rabring7), has been identified as a highly expressed protein in breast cancer cells and tissues. Later, it has been demonstrated that RNF115 catalyzes ubiquitination of a series of proteins to modulate a number of signaling pathways, and thereby regulates viral infections, autoimmunity, cell proliferation and death and tumorigenesis. In this review, we introduce the identification, expression and activity regulation of RNF115, summarize the substrates and functions of RNF115 in different pathways, and discuss the roles of RNF115 as a biomarker or therapeutic target in diseases.
Keywords: DNA damage repair; RNF115; antiviral immunity; autoimmunity; cancers.
Copyright © 2022 Wang, Liuyu and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

