Gene Therapy: Will the Promise of Optimizing Lung Allografts Become Reality?
- PMID: 35844566
- PMCID: PMC9283701
- DOI: 10.3389/fimmu.2022.931524
Gene Therapy: Will the Promise of Optimizing Lung Allografts Become Reality?
Abstract
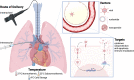
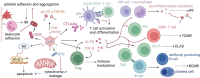
Lung transplantation is the definitive therapy for patients living with end-stage lung disease. Despite significant progress made in the field, graft survival remains the lowest of all solid organ transplants. Additionally, the lung has among the lowest of organ utilization rates-among eligible donors, only 22% of lungs from multi-organ donors were transplanted in 2019. Novel strategies are needed to rehabilitate marginal organs and improve graft survival. Gene therapy is one promising strategy in optimizing donor allografts. Over-expression or inhibition of specific genes can be achieved to target various pathways of graft injury, including ischemic-reperfusion injuries, humoral or cellular rejection, and chronic lung allograft dysfunction. Experiments in animal models have historically utilized adenovirus-based vectors and the majority of literature in lung transplantation has focused on overexpression of IL-10. Although several strategies were shown to prevent rejection and prolong graft survival in preclinical models, none have led to clinical translation. The past decade has seen a renaissance in the field of gene therapy and two AAV-based in vivo gene therapies are now FDA-approved for clinical use. Concurrently, normothermic ex vivo machine perfusion technology has emerged as an alternative to traditional static cold storage. This preservation method keeps organs physiologically active during storage and thus potentially offers a platform for gene therapy. This review will explore the advantages and disadvantages of various gene therapy modalities, review various candidate genes implicated in various stages of allograft injury and summarize the recent efforts in optimizing donor lungs using gene therapy.
Keywords: Adeno-associated viral vector (AAV vector); Adenoviral (Ad) vector; gene therapy; lung transplant; viral vector.
Copyright © 2022 Gao, DeLaura, Anwar, Kesseli, Kahan, Abraham, Asokan, Barbas and Hartwig.
Conflict of interest statement
MH consults for Bridge to Life, Paragonix, Medtronic, and Intuitive Surgical and has research funding from Cystic Fibrosis Foundation, Biomedinnovations, and Transmedics. AA is co-founder and director at StrideBio and TorqueBio. He is also an advisor to Kriya Therapeutics, Atsena Therapeutics, Ring Therapeutics and Mammoth Bio. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Chambers DC, Cherikh WS, Harhay MO, Hayes D, Jr., Hsich E, Khush KK, et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-Sixth Adult Lung and Heart-Lung Transplantation Report-2019; Focus Theme: Donor and Recipient Size Match. J Heart Lung Transplant (2019) 38:1042–55. doi: 10.1016/j.healun.2019.08.001 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

