Effects of dietary protein level on small intestinal morphology, occludin protein, and bacterial diversity in weaned piglets
- PMID: 35844902
- PMCID: PMC9281955
- DOI: 10.1002/fsn3.2828
Effects of dietary protein level on small intestinal morphology, occludin protein, and bacterial diversity in weaned piglets
Abstract
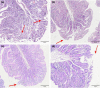
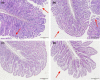
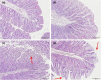
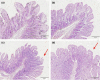
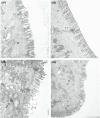
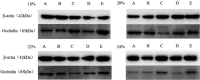
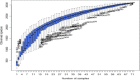
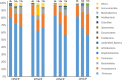
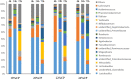
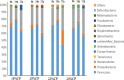
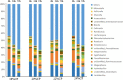
Due to the physiological characteristics of piglets, the morphological structure and function of the small intestinal mucosa change after weaning, which easily leads to diarrhea in piglets. The aim of this study was to investigate effects of crude protein (CP) levels on small intestinal morphology, occludin protein expression, and intestinal bacteria diversity in weaned piglets. Ninety-six weaned piglets (25 days of age) were randomly divided into four groups and fed diets containing 18%, 20%, 22%, and 24% protein. At 6, 24, 48, 72, and 96 h, changes in mucosal morphological structure, occludin mRNA, and protein expression and in the localization of occludin in jejunal and ileal tissues were evaluated. At 6, 24, and 72 h, changes in bacterial diversity and number of the ileal and colonic contents were analyzed. Results showed that structures of the jejunum and the ileum of piglets in the 20% CP group were intact. The expression of occludin mRNA and protein in the small intestine of piglets in the 20% CP group were significantly higher than those in the other groups. As the CP level increased, the number of pathogens, such as Clostridium difficile and Escherichia coli, in the intestine increased, while the number of beneficial bacteria, such as Lactobacillus, Bifidobacterium, and Roseburia, decreased. It is concluded that maintaining the CP level at 20% is beneficial to maintaining the small intestinal mucosal barrier and its absorption function, reducing the occurrence of diarrhea, and facilitating the growth and development of piglets.
Keywords: crude protein levels; diversity of intestinal bacteria; morphology and structure of small intestine; occludin protein.
© 2022 The Authors. Food Science & Nutrition published by Wiley Periodicals LLC.
Conflict of interest statement
The authors declare that they do not have any conflict of interest.
Figures
























References
-
- Caporaso, J. G. , Kuczynski, J. , Stombaugh, J. , Bittinger, K. , Bushman, F. D. , Costello, E. K. , Fierer, N. , Peña, A. G. , Goodrich, J. K. , Gordon, J. I. , Huttley, G. A. , Kelley, S. T. , Knights, D. , Koenig, J. E. , Ley, R. E. , Lozupone, C. A. , McDonald, D. , Muegge, B. D. , Pirrung, M. , … Knight, R. (2010). QIIME allows analysis of high‐throughput community sequencing data. Nature Methods, 7(5), 335–336. 10.1038/nmeth.f.303 - DOI - PMC - PubMed
-
- Dong, Y. , Fan, H. , Yao, S. , Wang, Y. , Yao, R. , Kong, X. , & Deng, J. (2019). Effects of dietary crude protein level on digestive capacity, colonic ammonia nitrogen content and inflammatory injury in weaned piglets. Chinese Journal of Animal Nutrition, 31(08), 3561–3570.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

