Effect of micro-arc oxidation surface modification of 3D-printed porous titanium alloys on biological properties
- PMID: 35845487
- PMCID: PMC9279779
- DOI: 10.21037/atm-22-2536
Effect of micro-arc oxidation surface modification of 3D-printed porous titanium alloys on biological properties
Abstract
Background: Three-dimensional (3D) printing technology has been widely used in orthopedics; however, it is still limited to the change of macroscopic structures. In order to further improve the biological properties of 3D-printed porous titanium scaffolds, this study introduced micro-arc oxidation (MAO) technology to modify the surface of porous titanium scaffolds and construct bioactive coatings on the surface of porous titanium scaffolds to improve the biocompatibility and osseointegration ability of the material.
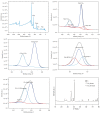
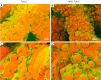
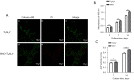
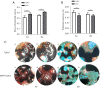
Methods: For in vitro experiments, human bone marrow stem cells (hBMSCs) were seeded onto untreated scaffolds (control group) and MAO-treated scaffolds (experimental group). After 24 h of co-culture, cytotoxicity was observed using live/dead staining, and cell/scaffold constructs were retrieved and processed for the assessment of cell morphology by using scanning electron microscopy (SEM). Cell proliferation was detected using the Cell Counting Kit-8 (CCK-8) assay after 3, 7, and 14 days of co-culture. The levels of alkaline phosphatase (ALP) in the cell supernatant were detected after 7 and 14 days of co-culture. For in vivo experiments, micro-computed tomography (micro-CT) and Masson Goldner's staining were used to evaluate bone ingrowth and osseointegration at 4 and 8 weeks postoperatively.
Results: In vitro experiment results confirmed that the two groups of scaffolds were non-cytotoxic and the cell adhesion status on the MAO-treated scaffolds was better. Over time, cell proliferation and ALP levels were higher in the MAO-treated group than in the untreated scaffolds. In the in vivo experiments, the MAO-treated scaffolds showed better bone ingrowth and osseointegration than the untreated group at different time points.
Conclusions: The MAO-treated porous titanium scaffold formed a uniform and dense bioactive coating on the surface, which was more conducive to cell adhesion, proliferation, and differentiation and showed better osseointegration and bone ingrowth in vivo.
Keywords: Three-dimensional printing (3D printing); biocompatibility; micro-arc oxidation (MAO); osseointegration.
2022 Annals of Translational Medicine. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://atm.amegroups.com/article/view/10.21037/atm-22-2536/coif). CW is from Beijing AK Medical Co., Ltd. The other authors have no conflicts of interest to declare.
Figures



Similar articles
-
Micro-arc oxidation (MAO) and its potential for improving the performance of titanium implants in biomedical applications.Front Bioeng Biotechnol. 2023 Nov 7;11:1282590. doi: 10.3389/fbioe.2023.1282590. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 38026886 Free PMC article. Review.
-
Multidynamic Osteogenic Differentiation by Effective Polydopamine Micro-Arc Oxide Manipulations.Int J Nanomedicine. 2022 Oct 11;17:4773-4790. doi: 10.2147/IJN.S378387. eCollection 2022. Int J Nanomedicine. 2022. PMID: 36246934 Free PMC article.
-
Early osseointegration of micro-arc oxidation coated titanium alloy implants containing Ag: a histomorphometric study.BMC Oral Health. 2022 Dec 22;22(1):628. doi: 10.1186/s12903-022-02673-6. BMC Oral Health. 2022. PMID: 36550526 Free PMC article.
-
Tailored Surface Treatment of 3D Printed Porous Ti6Al4V by Microarc Oxidation for Enhanced Osseointegration via Optimized Bone In-Growth Patterns and Interlocked Bone/Implant Interface.ACS Appl Mater Interfaces. 2016 Jul 20;8(28):17964-75. doi: 10.1021/acsami.6b05893. Epub 2016 Jul 5. ACS Appl Mater Interfaces. 2016. PMID: 27341499
-
Porous construction and surface modification of titanium-based materials for osteogenesis: A review.Front Bioeng Biotechnol. 2022 Aug 25;10:973297. doi: 10.3389/fbioe.2022.973297. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36091459 Free PMC article. Review.
Cited by
-
Influence of Ultrasound on the Characteristics of CaP Coatings Generated Via the Micro-arc Oxidation Process in Relation to Biomedical Engineering.ACS Biomater Sci Eng. 2024 Apr 8;10(4):2100-2115. doi: 10.1021/acsbiomaterials.3c01433. Epub 2024 Mar 19. ACS Biomater Sci Eng. 2024. PMID: 38502729 Free PMC article.
-
Remote Eradication of Bacteria on Orthopedic Implants via Delayed Delivery of Polycaprolactone Stabilized Polyvinylpyrrolidone Iodine.J Funct Biomater. 2022 Oct 19;13(4):195. doi: 10.3390/jfb13040195. J Funct Biomater. 2022. PMID: 36278664 Free PMC article.
-
Micro-arc oxidation (MAO) and its potential for improving the performance of titanium implants in biomedical applications.Front Bioeng Biotechnol. 2023 Nov 7;11:1282590. doi: 10.3389/fbioe.2023.1282590. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 38026886 Free PMC article. Review.
-
A Facile Surface Modification Strategy for Antibody Immobilization on 3D-Printed Surfaces.Biosensors (Basel). 2025 Mar 25;15(4):211. doi: 10.3390/bios15040211. Biosensors (Basel). 2025. PMID: 40277525 Free PMC article.
References
-
- Geetha M, Singh AK, Asokamani R, et al. Ti based biomaterials, the ultimate choice for orthopaedic implants–a review. Prog Mater Sci 2009;54:397-425. 10.1016/j.pmatsci.2008.06.004 - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
