New Enclosure for in vivo Medical Imaging of Zebrafish With Vital Signs Monitoring
- PMID: 35846002
- PMCID: PMC9278279
- DOI: 10.3389/fphys.2022.906110
New Enclosure for in vivo Medical Imaging of Zebrafish With Vital Signs Monitoring
Abstract
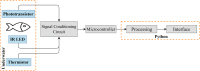
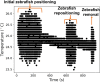
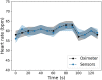
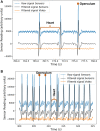
Lately, the use of zebrafish has gained increased interest in the scientific community as an animal model in preclinical research. However, there is a lack of in vivo imaging tools that ensure animal welfare during acquisition procedures. The use of functional imaging techniques, like Positron Emission Tomography (PET), in zebrafish is limited since it requires the animal to be alive, representing a higher instrumentation complexity when compared to morphological imaging systems. In the present work, a new zebrafish enclosure was developed to acquire in vivo images while monitoring the animal's welfare through its heartbeat. The temperature, dissolved oxygen, and pH range in a closed aquatic environment were tested to ensure that the conditions stay suitable for animal welfare during image acquisitions. The developed system, based on an enclosure with a bed and heartbeat sensors, was tested under controlled conditions in anesthetized fishes. Since the anesthetized zebrafish do not affect the water quality over time, there is no need to incorporate water circulation for the expected time of PET exams (about 30 min). The range of values obtained for the zebrafish heart rate was 88-127 bpm. The developed system has shown promising results regarding the zebrafish's heart rate while keeping the fish still during the long imaging exams. The zebrafish enclosure ensures the animal's well-being during the acquisition of in vivo images in different modalities (PET, Computer Tomography, Magnetic Resonance Imaging), contributing substantially to the preclinical research.
Keywords: Zebrafish physiology; non-invasive sensors; small animal imaging; vital signs monitoring; zebrafish heart rate.
Copyright © 2022 Magalhães, Correia, Oliveira, Encarnação, Domingues, Veloso and Silva.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







References
-
- Avdesh A., Chen M., Martin-Iverson M. T., Mondal A., Ong D., Rainey-Smith S., et al. (2012). Regular Care and Maintenance of a Zebrafish (Danio rerio) Laboratory: An Introduction. JoVE 69, 1–8. 10.3791/4196 PubMed Abstract | 10.3791/4196 | Google Scholar - DOI - PMC - PubMed
-
- Barbazuk W. B., Korf I., Kadavi C., Heyen J., Tate S., Wun E., et al. (2000). The Syntenic Relationship of the Zebrafish and Human Genomes. Genome Res. 10 (9), 1351–1358. 10.1101/gr.144700 PubMed Abstract | 10.1101/gr.144700 | Google Scholar - DOI - PMC - PubMed
-
- Browning Z. S. (2013). Using Advanced Imaging to Study Fish. Google Scholar
-
- Bufkin K., Leevy M. (2015). Multimodal Imaging Trials with Zebrafish Specimens. Winthrop McNair Res. Bull. 1 (3), 1–5. Google Scholar
-
- Collymore C., Tolwani A., Lieggi C., Rasmussen S. (2014). Efficacy and Safety of 5 Anesthetics in Adult Zebrafish (Danio rerio). J. Am. Assoc. Lab. Anim. Sci. 53 (2), 198–203. PubMed Abstract | Google Scholar - PMC - PubMed
LinkOut - more resources
Full Text Sources

