Smartphone Application for Celiac Patients: Assessing Its Effect on Gastrointestinal Symptoms in a Randomized Controlled Clinical Trial
- PMID: 35846977
- PMCID: PMC9286948
- DOI: 10.1155/2022/8027532
Smartphone Application for Celiac Patients: Assessing Its Effect on Gastrointestinal Symptoms in a Randomized Controlled Clinical Trial
Abstract
Introduction: Considering the lack of inclusive Persian application for celiac patients that covers all aspects of the GFD, we developed a Persian-language application for patients with CD and assessed the effectiveness of a three-month educational intervention delivered via smartphone application compared with standard care on gastrointestinal symptom rating scale (GSRS) score in patients with celiac disease.
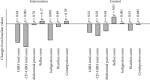
Methods: In the present parallel randomized controlled clinical trial, 60 patients with CD were assigned randomly to receive education through a smartphone application (n = 30) or conventional clinical education (n = 30). The patients were asked to use it for getting the required information for three months. We assessed the gastrointestinal symptoms using the gastrointestinal symptom rating scale (GSRS) questionnaire at baseline and three months after interventions. The GSRS total score, celiac disease GSRS (CD-GSRS) score, abdominal pain, reflux, diarrhea, constipation, and indigestion scores were calculated.
Results: Out of 60 randomized patients, 58 patients completed the study. In comparison to baseline, the mean score of CD-GSRS score (p = 0.001), and indigestion subscore (p < 0.001) were significantly decreased in the intervention group. The results of the between-group comparisons showed that there was a significant difference between the two groups only in the mean score of indigestion (p = 0.002).
Conclusion: According to the results, using a smartphone application for providing information to patients with celiac disease had a significant positive effect on indigestion symptoms compared with routine clinic education. Trial Registration. This trial is registered with the Iranian registry of clinical trials (IRCT code: IRCT20170117032004N2; trial registry date: 2019.6.26).
Copyright © 2022 Zeinab Nikniaz et al.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
The effect of group-based education on gastrointestinal symptoms and quality of life in patients with celiac disease: randomized controlled clinical trial.BMC Gastroenterol. 2022 Jan 11;22(1):18. doi: 10.1186/s12876-022-02096-1. BMC Gastroenterol. 2022. PMID: 35016615 Free PMC article. Clinical Trial.
-
Development and effectiveness assessment of a Persian-language smartphone application for celiac patients: A randomized controlled clinical trial.Patient Educ Couns. 2021 Feb;104(2):337-342. doi: 10.1016/j.pec.2020.08.014. Epub 2020 Aug 18. Patient Educ Couns. 2021. PMID: 32843265 Clinical Trial.
-
Beneficial Effects of a Low-Nickel Diet on Relapsing IBS-Like and Extraintestinal Symptoms of Celiac Patients during a Proper Gluten-Free Diet: Nickel Allergic Contact Mucositis in Suspected Non-Responsive Celiac Disease.Nutrients. 2020 Jul 29;12(8):2277. doi: 10.3390/nu12082277. Nutrients. 2020. PMID: 32751300 Free PMC article.
-
A Low FODMAP Diet Reduces Symptoms in Treated Celiac Patients With Ongoing Symptoms-A Randomized Controlled Trial.Clin Gastroenterol Hepatol. 2022 Oct;20(10):2258-2266.e3. doi: 10.1016/j.cgh.2022.01.011. Epub 2022 Jan 17. Clin Gastroenterol Hepatol. 2022. PMID: 35051648 Clinical Trial.
-
Larazotide acetate for treatment of celiac disease: A systematic review and meta-analysis of randomized controlled trials.Clin Res Hepatol Gastroenterol. 2022 Jan;46(1):101782. doi: 10.1016/j.clinre.2021.101782. Epub 2021 Jul 31. Clin Res Hepatol Gastroenterol. 2022. PMID: 34339872
Cited by
-
Follow-Up of Celiac Disease in Adults: "When, What, Who, and Where".Nutrients. 2023 Apr 24;15(9):2048. doi: 10.3390/nu15092048. Nutrients. 2023. PMID: 37432208 Free PMC article. Review.
-
Celiac Disease Course of Eritrean Asylum-Seeker Children Living in Israel Compared to Native Israeli Children.Acta Paediatr. 2025 Sep;114(9):2373-2381. doi: 10.1111/apa.70124. Epub 2025 May 7. Acta Paediatr. 2025. PMID: 40342135 Free PMC article.
-
Leveraging telemedicine in gastroenterology and hepatology: a narrative review.Mhealth. 2023 Oct 16;9:36. doi: 10.21037/mhealth-23-27. eCollection 2023. Mhealth. 2023. PMID: 38023778 Free PMC article. Review.
-
Telemedicine and the standard of care: a call for a new approach?Front Public Health. 2023 May 4;11:1184971. doi: 10.3389/fpubh.2023.1184971. eCollection 2023. Front Public Health. 2023. PMID: 37213629 Free PMC article.
-
Enhancing Self-Care Education in Celiac Disease Through Mobile Health: A Systematic Review.Health Sci Rep. 2025 Aug 6;8(8):e71086. doi: 10.1002/hsr2.71086. eCollection 2025 Aug. Health Sci Rep. 2025. PMID: 40777764 Free PMC article. Review.
References
-
- Akbari Namvar Z., Mahdavi R., Shirmohammadi M., Nikniaz Z. The effect of group-based education on knowledge and adherence to a gluten-free diet in patients with celiac disease: randomized controlled clinical trial. International Journal of Behavioral Medicine . 2021;28(5):583–590. doi: 10.1007/s12529-020-09949-7. - DOI - PubMed
LinkOut - more resources
Full Text Sources