Losac and Lopap Recombinant Proteins from Lonomia obliqua Bristles Positively Modulate the Myoblast Proliferation Process
- PMID: 35847970
- PMCID: PMC9280836
- DOI: 10.3389/fmolb.2022.904737
Losac and Lopap Recombinant Proteins from Lonomia obliqua Bristles Positively Modulate the Myoblast Proliferation Process
Abstract
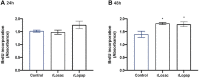
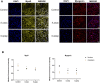
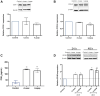
The pursuit of better therapies for disorders creating deficiencies in skeletal muscle regeneration is in progress, and several biotoxins are used in skeletal muscle research. Since recombinant proteins derived from Lonomia obliqua bristles, recombinant Lonomia obliqua Stuart-factor activator (rLosac) and recombinant Lonomia obliqua prothrombin activator protease (rLopap) act as cytoprotective agents and promote cell survival, we hypothesize that both rLosac and rLopap favour the skeletal muscle regeneration process. In the present work, we investigate the ability of these recombinant proteins rLosac and rLopap to modulate the production of key mediators of the myogenic process. The expression of myogenic regulatory factors (MRFs), cell proliferation, the production of prostaglandin E2 (PGE2) and the protein expression of cyclooxygenases COX-1 and COX-2 were evaluated in C2C12 mouse myoblasts pre-treated with rLosac and rLopap. We found an increased proliferation of myoblasts, stimulated by both recombinant proteins. Moreover, these proteins modulated PGE2 release and MRFs activities. We also found an increased expression of the EP4 receptor in the proliferative phase of C2C12 cells, suggesting the involvement of this receptor in the effects of PGE2 in these cells. Moreover, the recombinant proteins inhibited the release of IL-6 and PGE2, which is induced by an inflammatory stimulus by IL-1β. This work reveals rLopap and rLosac as promising proteins to modulate processes involving tissue regeneration as occurs during skeletal muscle injury.
Keywords: differentiation; inflammation; myoblast; proliferation; prostaglandin E2.
Copyright © 2022 Alvarez, Alvarez-Flores, DeOcesano-Pereira, Goldfeder, Chudzinski-Tavassi, Moreira and Teixeira.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Exploring new molecules and activities from Lonomia obliqua caterpillars.Pathophysiol Haemost Thromb. 2005;34(4-5):228-33. doi: 10.1159/000092429. Pathophysiol Haemost Thromb. 2005. PMID: 16707933 Review.
-
Losac, the first hemolin that exhibits procogulant activity through selective factor X proteolytic activation.J Biol Chem. 2011 Mar 4;286(9):6918-28. doi: 10.1074/jbc.M110.167718. Epub 2010 Dec 22. J Biol Chem. 2011. PMID: 21177860 Free PMC article.
-
Lopap: a non-inflammatory and cytoprotective molecule in neutrophils and endothelial cells.Toxicon. 2009 May;53(6):652-9. doi: 10.1016/j.toxicon.2009.01.031. Toxicon. 2009. PMID: 19673080
-
Losac, a factor X activator from Lonomia obliqua bristle extract: its role in the pathophysiological mechanisms and cell survival.Biochem Biophys Res Commun. 2006 May 19;343(4):1216-23. doi: 10.1016/j.bbrc.2006.03.068. Epub 2006 Mar 20. Biochem Biophys Res Commun. 2006. PMID: 16597435
-
Regulation of Skeletal Muscle Myoblast Differentiation and Proliferation by Pannexins.Adv Exp Med Biol. 2017;925:57-73. doi: 10.1007/5584_2016_53. Adv Exp Med Biol. 2017. PMID: 27518505 Review.
References
-
- Alvarez Flores M. P., Fritzen M., Reis C. V., Chudzinski-Tavassi A. M. (2006). Losac, a Factor X Activator from Lonomia Obliqua Bristle Extract: Its Role in the Pathophysiological Mechanisms and Cell Survival. Biochem. Biophysical Res. Commun. 343, 1216–1223. 10.1016/j.bbrc.2006.03.068 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

