Isothiourea-Catalyzed [2 + 2] Cycloaddition of C(1)-Ammonium Enolates and N-Alkyl Isatins
- PMID: 35848722
- PMCID: PMC9490795
- DOI: 10.1021/acs.orglett.2c02170
Isothiourea-Catalyzed [2 + 2] Cycloaddition of C(1)-Ammonium Enolates and N-Alkyl Isatins
Abstract
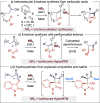
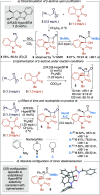
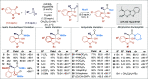
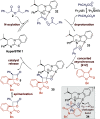
Enantioselective [2 + 2] cycloaddition of C(1)-ammonium enolates generated catalytically using the isothiourea HyperBTM with N-alkyl isatins gives spirocyclic β-lactones. In situ ring opening with an amine nucleophile generates isolable highly enantioenriched products in up to 92:8 dr and in >99:1 er.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
-
-
For selected reviews, see:
- Mukherjee S.; Biju A. Recent advances in the organocatalytic enantioselective synthesis of functionalized β-lactones. Chem.—Asian J. 2018, 13, 2333. 10.1002/asia.201800902. - DOI - PubMed
- Romo D.; Tennyson R. L.; Wang Y. β-Lactones as intermediates for natural product total synthesis and new transformations. Heterocycles 2004, 64, 605–658. 10.3987/REV-04-SR(P)3. - DOI
-
-
-
For selected examples see;
- Pommier A.; Pons J.-M. The synthesis of natural 2-oxetanones. Synthesis 1995, 1995, 729–744. 10.1055/s-1995-4011. - DOI
- Lowe C.; Vederas J. C. Naturally occurring β-lactones: Occurrence, syntheses and properties. A review. Org. Prep. Proced. Int. 1995, 27, 305–346. 10.1080/00304949509458466. - DOI
- Yang H. W.; Romo D. Methods for the synthesis of optically active β-lactones. Tetrahedron 1999, 55, 6403–6434. 10.1016/S0040-4020(99)00185-4. - DOI
- Böttcher T.; Sieber S. A. β-Lactams and β-lactones as activity-based probes in chemical biology. Med. Chem. Comm. 2012, 3, 408–417. 10.1039/c2md00275b. - DOI
-
-
-
For reviews see:
- Schneider C. Catalytic, enantioselective syntheses of β-lactones-versatile synthetic building blocks in organic chemistry. Angew. Chem., Int. Ed. 2002, 41, 744–746. 10.1002/1521-3773(20020301)41:5<744::AID-ANIE744>3.0.CO;2-V. - DOI - PubMed
- Douglas J.; Morrill L. C.; Richmond E.; Smith A. D. In Methods and Applications of Cycloaddition Reactions in Organic Syntheses; Nishiwaki N., Ed.; Wiley: Hoboken NJ, 2014; ch. 3, pp 89–114.
- Van K. N.; Morrill L. C.; Smith A. D.; Romo D. In Lewis Base Catalysis in Organic Synthesis; Vedejs E., Denmark S. E., Eds.; Wiley-VCH: Weinheim, 2016; Vol. 2, ch. 13, pp 527–653.
-
-
- Wynberg H.; Staring E. G. J. Asymmetric synthesis of (S)- and (R)-malic acid from ketene and chloral. J. Am. Chem. Soc. 1982, 104, 166–168. 10.1021/ja00365a030. - DOI
- Wynberg H.; Staring E. G. J. Catalytic asymmetric synthesis of chiral 4-substituted oxetanones. J. Org. Chem. 1985, 50, 1977–1979. 10.1021/jo00211a039. - DOI
- Wynberg H.; Staring E. G. J. The absolute configuration of 4-(trichloromethyl)oxetan-2-one; a case of double anchimeric assistance with inversion. J. Chem. Soc., Chem. Commun. 1984, 1181–1182. 10.1039/c39840001181. - DOI
- Calter M. A. J. Org. Chem. 1996, 61, 8006–8007. 10.1021/jo961721c. - DOI - PubMed
- Tennyson R.; Romo D. Use of In Situ Generated Ketene in the Wynberg β-Lactone Synthesis: New Transformations of the Dichlorinated β-Lactone Products. J. Org. Chem. 2000, 65, 7248–7252. 10.1021/jo001010l. - DOI - PubMed
- Calter M. A.; Orr R. K.; Song W. Org. Lett. 2003, 5, 4745–4748. 10.1021/ol0359517. - DOI - PubMed
- Zhu C.; Shen X.; Nelson S. G. J. Am. Chem. Soc. 2004, 126, 5352–5353. 10.1021/ja0492900. - DOI - PubMed
- Calter M. A.; Tretyak O. A.; Flaschenriem C. Org. Lett. 2005, 7, 1809–1812. 10.1021/ol050411q. - DOI - PubMed
- Green M. E.; Rech J. C.; Floreancig P. E. Angew. Chem., Int. Ed. 2008, 47, 7317–7320. 10.1002/anie.200802548. - DOI - PMC - PubMed
- Jiang X.; Fu C.; Ma S. Chem.—Eur. J. 2008, 14, 9656–9664. 10.1002/chem.200801363. - DOI - PubMed
- Chandra B.; Fu D.; Nelson S. G. Angew. Chem., Int. Ed. 2010, 49, 2591–2594. 10.1002/anie.200906245. - DOI - PubMed
- Vargo T. R.; Hale J. S.; Nelson S. G. Angew. Chem., Int. Ed. 2010, 49, 8678–8681. 10.1002/anie.201004925. - DOI - PubMed
- Wan S.; Wu F.; Rech J. C.; Green M. E.; Balachandran R.; Horne W. S.; Day B. W.; Floreancig P. E. J. Am. Chem. Soc. 2011, 133, 16668–16679. 10.1021/ja207331m. - DOI - PubMed
-
- Wilson J. E.; Fu G. C. Asymmetric synthesis of highly substituted β-lactones by nucleophile-catalyzed [2 + 2] cycloadditions of disubstituted ketenes with aldehydes. Angew. Chem., Int. Ed. 2004, 43, 6358–6360. 10.1002/anie.200460698. - DOI - PubMed
- Zhu C.; Shen X.; Nelson S. G. Cinchona alkaloid-lewis acid catalyst systems for enantioselective ketene–aldehyde cycloadditions. J. Am. Chem. Soc. 2004, 126, 5352–5353. 10.1021/ja0492900. - DOI - PubMed
-
For a selected NHC-catalyzed process, see:
- He L.; Lv H.; Zhang Y.; Ye S. Formal cycloaddition of disubstituted ketenes with 2-oxoaldehydes catalyzed by chiral N-heterocyclic carbenes. J. Org. Chem. 2008, 73, 8101–8103. 10.1021/jo801494f. - DOI - PubMed
- Douglas J.; Taylor J. E.; Churchill G.; Slawin A. M. Z.; Smith A. D. NHC-Promoted asymmetric β-lactone formation from arylalkylketenes and electron-deficient benzaldehydes or pyridinecarboxaldehydes. J. Org. Chem. 2013, 78, 3925–3938. 10.1021/jo4003079. - DOI - PubMed
- Davies A. T.; Slawin A. M. Z.; Smith A. D. Enantioselective NHC-Catalyzed Redox [2 + 2] Cycloadditions with Perfluoroketones; A Route to Fluorinated Oxetanes. Chem.—Eur. J. 2015, 21, 18944–18948. 10.1002/chem.201504256. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

