Impact of the Clinical Trials Act 2018 on clinical trial activity in Japan from 2018 to 2020: a retrospective database study using new and conventional Japanese registries
- PMID: 35851007
- PMCID: PMC9297204
- DOI: 10.1136/bmjopen-2021-059092
Impact of the Clinical Trials Act 2018 on clinical trial activity in Japan from 2018 to 2020: a retrospective database study using new and conventional Japanese registries
Abstract
Objective: To clarify the impact of Japan's Clinical Trials Act (CTA), which was enacted in April 2018, on subsequent clinical trial activity through an analysis of Japanese registry data.
Design: Retrospective database study.
Setting: We extracted information on clinical intervention studies registered between 1 April 2018 and 30 September 2020 in the conventional University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR) and the new Japan Registry of Clinical Trials (jRCT). We collected and analysed information on registration dates, intervention types, funding, secondary sponsors and use of designated staff in multidisciplinary roles (research planning support, research administration, data management, statistical analysis, monitoring and auditing). The temporal trends in clinical trial activity after CTA enactment were examined.
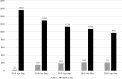
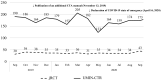
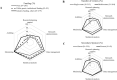
Results: A total of 577 CTA-compliant specified clinical trials (ie, studies funded by pharmaceutical companies or studies evaluating the efficacy and safety of off-label drugs or devices in humans) were registered in the jRCT. During the same period, 5068 clinical trials were registered in the UMIN-CTR. The number of specific clinical trials increased immediately after the implementation of the CTA and stabilised in late 2019, whereas the number of clinical trials registered in the UMIN-CTR generally declined over time. Specified clinical trials that received industry funding and public grants were more likely to use designated staff in multidisciplinary roles.
Conclusions: The implementation of the CTA has not reduced the number of specified clinical trials, but has reduced the total number of intervention trials. The use of designated staff in multidisciplinary roles is associated with funding, secondary sponsors and multicentre studies. It was inferred that funding was needed to establish research infrastructure systems that support high-quality research.
Keywords: clinical governance; clinical trials; health policy.
© Author(s) (or their employer(s)) 2022. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: HT and OT are employees of Daiichi Sankyo. HT is also an employee of the Cancer Institute Hospital of the Japanese Foundation for Cancer Research. KM is an employee of Daiichi Sankyo RD Novare.
Figures

Similar articles
-
Review of the registration of clinical trials in UMIN-CTR from 2 June 2005 to 1 June 2010 - focus on Japan domestic, academic clinical trials.Trials. 2013 Oct 14;14:333. doi: 10.1186/1745-6215-14-333. Trials. 2013. PMID: 24124926 Free PMC article.
-
Publication of lung cancer clinical trials in the Japanese Clinical Trial Registry.Jpn J Clin Oncol. 2018 Nov 1;48(11):995-1000. doi: 10.1093/jjco/hyy131. Jpn J Clin Oncol. 2018. PMID: 30256970
-
Impact of the Clinical Trials Act on Noncommercial Clinical Research in Japan: An Interrupted Time-series Analysis.J Epidemiol. 2022 Jan 5;32(1):27-33. doi: 10.2188/jea.JE20210051. Epub 2021 Nov 25. J Epidemiol. 2022. PMID: 34690242 Free PMC article.
-
[Infrastructure for new drug development to treat muscular dystrophy: current status of patient registration (remudy)].Brain Nerve. 2011 Nov;63(11):1279-84. Brain Nerve. 2011. PMID: 22068481 Review. Japanese.
-
[Legislation of cancer registries in Japan- an outline of the national cancer registry].Gan To Kagaku Ryoho. 2015 Apr;42(4):389-93. Gan To Kagaku Ryoho. 2015. PMID: 25963684 Review. Japanese.
Cited by
-
East Asian perspective of responsible research and innovation in neurotechnology.IBRO Neurosci Rep. 2024 May 5;16:582-597. doi: 10.1016/j.ibneur.2024.04.009. eCollection 2024 Jun. IBRO Neurosci Rep. 2024. PMID: 38774060 Free PMC article.
-
Transitional dynamics in oncology clinical trials: evaluating the impact of Clinical Trials Act on cooperative groups.Jpn J Clin Oncol. 2024 Jul 7;54(7):748-752. doi: 10.1093/jjco/hyae034. Jpn J Clin Oncol. 2024. PMID: 38535873 Free PMC article.
References
-
- U.S. Department of Health & Human Services . Final rule delaying general compliance of revised common rule, 2018. Available: https://www.hhs.gov/ohrp/final-rule-delaying-general-compliance-revised-... [Accessed 9 Oct 2021].
-
- Rice TW. The historical, ethical, and legal background of human-subjects research. Respir Care 2008;53:1325–9. - PubMed
-
- Kurihara C. Reformation of clinical trial regulations in European Union (EU). Rinsyo Hyoka/Clinical Eval 2014;42:485–500.
-
- For life sciences and medical research involving human subjects ethical guideline. Available: https://www.lifescience.mext.go.jp/files/pdf/n2312_01.pdf
-
- Law on securing quality, efficacy and safety of pharmaceuticals, medical devices, regenerative and cellular therapy puroducts, gene therapy products, and cosmetics, 2015. Available: https://www.jiho.co.jp/Portals/0/ec/product/ebooks/book/47578/47578.pdf
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
