The Metabolic Landscape in Osteoarthritis
- PMID: 35855332
- PMCID: PMC9286923
- DOI: 10.14336/AD.2021.1228
The Metabolic Landscape in Osteoarthritis
Abstract
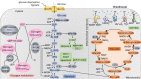
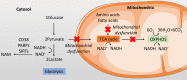
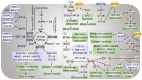
Articular cartilage function depends on the temporal and zonal distribution of coordinated metabolic regulation in chondrocytes. Emerging evidence shows the importance of cellular metabolism in the molecular control of the cartilage and its dysregulation in degenerative diseases like osteoarthritis (OA). Compared to most other tissues, chondrocytes are sparsely located in the extracellular matrix, lacking the typical proximity of neural, vascular, and lymphatic tissue. Making up under 5% of the total tissue weight of cartilage, chondrocytes have a relative deficiency of access to nutrients and oxygen, as well as limited pathways for metabolite removal. This makes cartilage a unique tissue with hypocellularity, prolonged metabolic rate, and tissue turnover. Studies in the past decade have shown that several pathways of central carbon metabolism are essential for cartilage homeostasis. Here, we summarised the literature findings on the role of cellular metabolism in determining the chondrocyte function and how this metabolic dysregulation led to cartilage aging in OA and provided an outlook on how the field may evolve in the coming years. Although the various energy metabolism pathways are inextricably linked with one another, for the purpose of this review, we initially endeavoured to examine them individually and in relative isolation. Subsequently, we comment on what is known regarding the integration and linked signalling pathways between these systems and the therapeutic opportunities for targeting OA metabolism.
Keywords: articular cartilage; chondrocyte; energy metabolism; glycolysis; osteoarthritis; oxidative phosphorylation.
copyright: © 2022 Wu et al.
Conflict of interest statement
Conflicts of interest The authors have no conflicts of interest to declare.
Figures


References
-
- Zheng L, Zhang Z, Sheng P, Mobasheri A (2020). The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis. Ageing Res Rev, 66:101249. - PubMed
-
- Oikonomou E, Tsioufis C, Tousoulis D (2021). Diabetes mellitus: a primary metabolic disturbance. Metabolomics underlying vascular responses to stress and ischemia? Clin Sci, 135:589-591. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
