Effect of Text Message Reminders and Vaccine Reservations on Adherence to a Health System COVID-19 Vaccination Policy: A Randomized Clinical Trial
- PMID: 35857327
- PMCID: PMC9301516
- DOI: 10.1001/jamanetworkopen.2022.22116
Effect of Text Message Reminders and Vaccine Reservations on Adherence to a Health System COVID-19 Vaccination Policy: A Randomized Clinical Trial
Abstract
Importance: Many organizations implemented COVID-19 vaccination requirements during the pandemic, but the best way to increase adherence to these policies is unknown.
Objective: To evaluate if behavioral nudges delivered through text messages could accelerate adherence to a health system's COVID-19 vaccination policy.
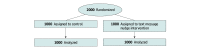
Design, setting, and participants: This randomized clinical trial was conducted within Ascension health system from October 11 to November 8, 2021. Participants included health system employees in the Midwest or South US who were not adherent with the vaccination policy 1 month before its deadline. Data were analyzed from November 17, 2021, to February 25, 2022.
Interventions: Participants were randomly assigned to control or to receive a text message intervention that stated a vaccine had been reserved for the participant, with a scheduled date for vaccination within a 2-week period. Participants could reschedule to a different date within the period or upload a copy of their vaccination card. Follow-up text message reminders were sent the day before and the day of the appointment.
Main outcomes and measures: The primary outcome was adherence to the health system's vaccination policy during the 2-week intervention. Secondary outcomes included time to vaccination during a 4-week follow-up period.
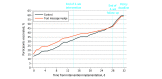
Results: The sample included 2000 participants (mean [SD] age, 36.4 [12.3] years; 1724 [86.2%] women), with 1000 participants randomized to the control group and 1000 participants randomized to the intervention group. Overall, there were 164 Hispanic participants (8.2%), 46 non-Hispanic Asian participants (2.3%), 202 non-Hispanic Black participants (10.1%), and 1418 non-Hispanic White participants (70.9%). By the end of the 2-week intervention, 363 participants in the text message nudge group (36.3%) and 318 participants in the control group (31.8%) were adherent with the vaccination policy, representing a significant increase of 4.9 (95% CI, 0.8 to 9.1) percentage points in adjusted analyses comparing the nudge group with the control group (P = .02). Among participants who became adherent by the end of the 4-week follow-up period, the text message nudge significantly reduced time to adherence by a mean of 2.4 (95% CI, 2.1 to 4.7) days (P < .001) and a median of 5.0 (95% CI, 2.5 to 7.7) days (P < .001) compared with the control group. At 4 weeks, overall vaccination adherence was no longer different between groups (control: 477 participants [47.7%]; intervention: 472 participants [47.2%]).
Conclusions and relevance: This randomized clinical trial found that a behavioral nudge delivered through text messages accelerated adherence to a health system's COVID-19 vaccination policy but did change overall adherence by the time of the policy deadline.
Trial registration: ClinicalTrials.gov Identifier: NCT05037201.
Conflict of interest statement
Figures
Similar articles
-
Effect of Text Messaging and Behavioral Interventions on COVID-19 Vaccination Uptake: A Randomized Clinical Trial.JAMA Netw Open. 2022 Jun 1;5(6):e2216649. doi: 10.1001/jamanetworkopen.2022.16649. JAMA Netw Open. 2022. PMID: 35696165 Free PMC article. Clinical Trial.
-
Effects of Ownership Text Message Wording and Reminders on Receipt of an Influenza Vaccination: A Randomized Clinical Trial.JAMA Netw Open. 2022 Feb 1;5(2):e2143388. doi: 10.1001/jamanetworkopen.2021.43388. JAMA Netw Open. 2022. PMID: 35175346 Free PMC article. Clinical Trial.
-
Short Message Service Reminder Nudge for Parents and Influenza Vaccination Uptake in Children and Adolescents With Special Risk Medical Conditions: The Flutext-4U Randomized Clinical Trial.JAMA Pediatr. 2023 Apr 1;177(4):337-344. doi: 10.1001/jamapediatrics.2022.6145. JAMA Pediatr. 2023. PMID: 36806893 Free PMC article. Clinical Trial.
-
Effectiveness of mobile phone text message reminder interventions to improve adherence to antiretroviral therapy among adolescents living with HIV: A systematic review and meta-analysis.PLoS One. 2021 Jul 22;16(7):e0254890. doi: 10.1371/journal.pone.0254890. eCollection 2021. PLoS One. 2021. PMID: 34293033 Free PMC article.
-
Mobile Telephone Text Messaging for Medication Adherence in Chronic Disease: A Meta-analysis.JAMA Intern Med. 2016 Mar;176(3):340-9. doi: 10.1001/jamainternmed.2015.7667. JAMA Intern Med. 2016. PMID: 26831740 Review.
Cited by
-
Era of Generalist Conversational Artificial Intelligence to Support Public Health Communications.J Med Internet Res. 2025 Jan 20;27:e69007. doi: 10.2196/69007. J Med Internet Res. 2025. PMID: 39832358 Free PMC article.
-
"How" Versus "Why" Messaging to Increase Uptake of Booster Vaccination Against COVID-19: Results of a Pragmatic Randomized Trial.J Gen Intern Med. 2024 Mar;39(4):611-618. doi: 10.1007/s11606-023-08492-x. Epub 2023 Nov 6. J Gen Intern Med. 2024. PMID: 37932539 Free PMC article. Clinical Trial.
-
Behaviorally Informed Text Message Nudges to Schedule COVID-19 Vaccinations: A Randomized Controlled Trial.J Gen Intern Med. 2025 Apr;40(5):1078-1084. doi: 10.1007/s11606-024-09170-2. Epub 2024 Nov 4. J Gen Intern Med. 2025. PMID: 39496851 Clinical Trial.
-
Nationwide demonstration of improved COVID-19 vaccination uptake through behavioural reminders.Nat Hum Behav. 2025 Jun;9(6):1237-1245. doi: 10.1038/s41562-025-02165-x. Epub 2025 Apr 9. Nat Hum Behav. 2025. PMID: 40204873 Clinical Trial.
-
Effectiveness of Text Messaging Nudging to Increase Coverage of Influenza Vaccination Among Older Adults in Norway (InfluSMS Study): Protocol for a Randomized Controlled Trial.JMIR Res Protoc. 2025 Feb 25;14:e63938. doi: 10.2196/63938. JMIR Res Protoc. 2025. PMID: 39998878 Free PMC article.
References
-
- Richardson S, Hirsch JS, Narasimhan M, et al. ; the Northwell COVID-19 Research Consortium . Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052-2059. doi:10.1001/jama.2020.6775 - DOI - PMC - PubMed