N-Acetylcysteine (NAC) in Schizophrenia Resistant to Clozapine: A Double-Blind, Randomized, Placebo-Controlled Trial Targeting Negative Symptoms
- PMID: 35857752
- PMCID: PMC9673271
- DOI: 10.1093/schbul/sbac065
N-Acetylcysteine (NAC) in Schizophrenia Resistant to Clozapine: A Double-Blind, Randomized, Placebo-Controlled Trial Targeting Negative Symptoms
Abstract
Background and hypothesis: Clozapine is the most effective antipsychotic for treatment-resistant schizophrenia, yet a significant proportion of individuals on clozapine continue to experience disabling symptoms, despite being treated with an adequate dose. There is a need for adjunct treatments to augment clozapine, notably for negative and cognitive symptoms. One such potential agent is the glutathione precursor N-acetylcysteine (NAC).
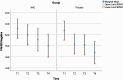
Study design: A randomized double-blind, multi-center, placebo-controlled trial for clozapine patients with enduring psychotic symptoms (n = 84) was undertaken to investigate the efficacy of adjunctive NAC (2 g daily) for negative symptoms, cognition and quality of life (QoL). Efficacy was assessed at 8, 24, and 52 weeks.
Study results: NAC did not significantly improve negative symptoms (P = .62), overall cognition (P = .71) or quality of life (Manchester quality of life: P = .11; Assessment of quality of life: P = .57) at any time point over a 1-year period of treatment. There were no differences in reported side effects between the groups (P = .26).
Conclusions: NAC did not significantly improve schizophrenia symptoms, cognition, or quality of life in treatment-resistant patients taking clozapine. This trial was registered with "Australian and New Zealand Clinical Trials" on the 30 May, 2016 (Registration Number: ACTRN12615001273572).
Keywords: cognition; depression; mental disorders; mental illness; neuroscience; psychiatry; quality of life.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Maryland Psychiatric Research Center.
Figures
References
-
- Lally J, MacCabe JH.. Antipsychotic medication in schizophrenia: a review. Br Med Bull. 2015;114(1):169–179. - PubMed
-
- Siskind D, Orr S, Sinha S, et al. Rates of treatment-resistant schizophrenia from first-episode cohorts: systematic review and meta-analysis. Br J Psychiatry. 2021;220(3):1–6. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical