Peripheral lymphocyte subset alteration in patients with COVID-19 having differential clinical manifestations
- PMID: 35859440
- PMCID: PMC9552374
- DOI: 10.4103/ijmr.IJMR_453_21
Peripheral lymphocyte subset alteration in patients with COVID-19 having differential clinical manifestations
Abstract
Background & objectives: The COVID-19 disease profile in Indian patients has been found to be different from the Western world. Changes in lymphocyte compartment have been correlated with disease course, illness severity and clinical outcome. This study was aimed to assess the peripheral lymphocyte phenotype and subset distribution in patients with COVID-19 disease from India with differential clinical manifestations.
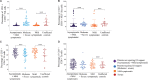
Methods: Percentages of peripheral lymphocyte subsets were measured by flow cytometry in hospitalized asymptomatic (n=53), mild symptomatic (n=36), moderate and severe (n=30) patients with SARS-CoV-2 infection, recovered individuals (n=40) and uninfected controls (n=56) from Pune, Maharashtra, India.
Results: Percentages of CD4+Th cells were significantly high in asymptomatic, mild symptomatic, moderate and severe patients and recovered individuals compared to controls. Percentages of Th memory (CD3+CD4+CD45RO+), Tc memory (CD3+CD8+CD45RO+) and B memory (CD19+CD27+) cells were significantly higher in the recovered group compared to both asymptomatic, mild symptomatic patient and uninfected control groups. NK cell (CD56+CD3-) percentages were comparable among moderate +severe patient and uninfected control groups.
Interpretation & conclusions: The observed lower CD4+Th cells in moderate+severe group requiring oxygen support compared to asymptomatic+mild symptomatic group not requiring oxygen support could be indicative of poor prognosis. Higher Th memory, Tc memory and B memory cells in the recovered group compared to mild symptomatic patient groups might be markers of recovery from mild infection; however, it remains to be established if the persistence of any of these cells could be considered as a correlate of protection.
Keywords: Asymptomatic; SARS-CoV-2; lymphocyte subset; mild symptomatic; moderate +severe patients; recovered individuals.
Conflict of interest statement
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
