Gene expression and involvement of signaling pathways during host-pathogen interplay in Orientia tsutsugamushi infection
- PMID: 35860421
- PMCID: PMC9295102
- DOI: 10.1007/s13205-022-03239-7
Gene expression and involvement of signaling pathways during host-pathogen interplay in Orientia tsutsugamushi infection
Abstract
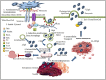
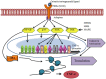
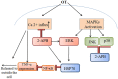
Scrub typhus is a neglected tropical disease that affects one-third of the world's population. The disease is caused by Orientia tsutsugamushi (OT), an obligate intracellular Gram-negative bacterium. OT efficiently escapes from the endosomal pathway after entering the host cell and replicates inside cytosol. OT infection promotes cellular autophagy, the autonomous defense mechanism unlike other bacteria. This study has discussed the bacterial invasion process through the extracellular matrix and the immune response activated by the bacterium within the hosts. Furthermore, we have emphasized the importance of extracellular matrix and their cross-talk with the immune cells, such as, macrophages, neutrophils, and dendritic cells followed by their inflammatory response. We have also put an insight into the host factors associated with signaling pathways during scrub typhus disease with a special focus on the OT-induced stress response, autophagy, apoptosis, and innate immunity. Multiple cytokines and chemokines play a significant role in activating different immune-related signaling pathways. Due to the presence of high antigenic diversity among strains, the signaling pathways during the host-pathogen interplay of OT with its host is very complicated. Thus, it hinders to mitigate the severity of the pandemic occurred by the respective pathogen. Our investigation will provide a useful guide to better understand the virulence and physiology of this intracellular pathogen which will lead towards a better therapeutic diagnosis and vaccine development.
Keywords: Gene expression; Host–pathogen interaction; Inflammatory response; Orientia tsutsugamushi.
© King Abdulaziz City for Science and Technology 2022.
Conflict of interest statement
Conflict of interestThe authors have no conflict of interest to declare.
Figures
Similar articles
-
Active escape of Orientia tsutsugamushi from cellular autophagy.Infect Immun. 2013 Feb;81(2):552-9. doi: 10.1128/IAI.00861-12. Epub 2012 Dec 10. Infect Immun. 2013. PMID: 23230293 Free PMC article.
-
Murine models of scrub typhus associated with host control of Orientia tsutsugamushi infection.PLoS Negl Trop Dis. 2017 Mar 10;11(3):e0005453. doi: 10.1371/journal.pntd.0005453. eCollection 2017 Mar. PLoS Negl Trop Dis. 2017. PMID: 28282373 Free PMC article.
-
CCR7/dendritic cell axis mediates early bacterial dissemination in Orientia tsutsugamushi-infected mice.Front Immunol. 2022 Dec 22;13:1061031. doi: 10.3389/fimmu.2022.1061031. eCollection 2022. Front Immunol. 2022. PMID: 36618364 Free PMC article.
-
Orientia tsutsugamushi: The dangerous yet neglected foe from the East.Int J Med Microbiol. 2021 Jan;311(1):151467. doi: 10.1016/j.ijmm.2020.151467. Epub 2020 Dec 9. Int J Med Microbiol. 2021. PMID: 33338890
-
Scrub Typhus Pathogenesis: Innate Immune Response and Lung Injury During Orientia tsutsugamushi Infection.Front Microbiol. 2019 Sep 6;10:2065. doi: 10.3389/fmicb.2019.02065. eCollection 2019. Front Microbiol. 2019. PMID: 31555249 Free PMC article. Review.
Cited by
-
Varicella-Zoster Virus Reactivation During the Incubation Period for Scrub Typhus: A Case Report.Am J Case Rep. 2022 Nov 21;23:e937559. doi: 10.12659/AJCR.937559. Am J Case Rep. 2022. PMID: 36409660 Free PMC article.
-
Epidemiological analysis and risk prediction of scrub typhus from 2006 to 2021 in Sichuan, China.Front Public Health. 2023 May 30;11:1177578. doi: 10.3389/fpubh.2023.1177578. eCollection 2023. Front Public Health. 2023. PMID: 37325301 Free PMC article. Review.
-
Development and implementation of a strategy for early diagnosis and management of scrub typhus: an emerging public health threat.Front Public Health. 2024 Apr 10;12:1347183. doi: 10.3389/fpubh.2024.1347183. eCollection 2024. Front Public Health. 2024. PMID: 38660358 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

