PLCβ2 Promotes VEGF-Induced Vascular Permeability
- PMID: 35861069
- PMCID: PMC9492642
- DOI: 10.1161/ATVBAHA.122.317645
PLCβ2 Promotes VEGF-Induced Vascular Permeability
Abstract
Background: Regulation of vascular permeability is critical to maintaining tissue metabolic homeostasis. VEGF (vascular endothelial growth factor) is a key stimulus of vascular permeability in acute and chronic diseases including ischemia reperfusion injury, sepsis, and cancer. Identification of novel regulators of vascular permeability would allow for the development of effective targeted therapeutics for patients with unmet medical need.
Methods: In vitro and in vivo models of VEGFA-induced vascular permeability, pathological permeability, quantitation of intracellular calcium release and cell entry, and phosphatidylinositol 4,5-bisphosphate levels were evaluated with and without modulation of PLC (phospholipase C) β2.
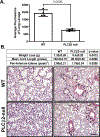
Results: Global knock-out of PLCβ2 in mice resulted in blockade of VEGFA-induced vascular permeability in vivo and transendothelial permeability in primary lung endothelial cells. Further work in an immortalized human microvascular cell line modulated with stable knockdown of PLCβ2 recapitulated the observations in the mouse model and primary cell assays. Additionally, loss of PLCβ2 limited both intracellular release and extracellular entry of calcium following VEGF stimulation as well as reduced basal and VEGFA-stimulated levels of phosphatidylinositol 4,5-bisphosphate compared to control cells. Finally, loss of PLCβ2 in both a hyperoxia-induced lung permeability model and a cardiac ischemia:reperfusion model resulted in improved animal outcomes when compared with wild-type controls.
Conclusions: The results implicate PLCβ2 as a key positive regulator of VEGF-induced vascular permeability through regulation of both calcium flux and phosphatidylinositol 4,5-bisphosphate levels at the cellular level. Targeting of PLCβ2 in a therapeutic setting may provide a novel approach to regulating vascular permeability in patients.
Keywords: homeostasis; ischemia; permeability; reperfusion; vascular endothelial growth factor A.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures
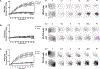




Comment in
-
Novel Target for Limiting VEGF-A (Vascular Endothelial Growth Factor A)-Induced Vascular Permeability.Arterioscler Thromb Vasc Biol. 2022 Oct;42(10):1242-1243. doi: 10.1161/ATVBAHA.122.318105. Epub 2022 Aug 25. Arterioscler Thromb Vasc Biol. 2022. PMID: 36004641 No abstract available.
References
-
- Goddard LM, Iruela-Arispe ML. Cellular and molecular regulation of vascular permeability. Thromb Haemostasis [Internet]. 2013;109:407–415. Available from: http://www.schattauer.de/en/magazine/subject-areas/journals-a-z/thrombos... - PMC - PubMed
-
- Stevens T, Garcia JGN, Shasby DM, Bhattacharya J, Malik AB. Mechanisms regulating endothelial cell barrier function. Am J Physiol-lung C [Internet]. 2000;279:419–L422. Available from: http://ajplung.physiology.org/cgi/content/abstract/279/3/L419 - PubMed
-
- Nagy J, Benjamin L, Zeng H, Dvorak A, Dvorak H. Vascular permeability, vascular hyperpermeability and angiogenesis. Angiogenesis [Internet]. 2008;11:109–119. Available from: https://www.ncbi.nlm.nih.gov/pubmed/18293091 - PMC - PubMed
-
- Lionetti V, Recchia FA, Ranieri VM. Overview of ventilator-induced lung injury mechanisms. Curr Opin Crit Care [Internet]. 2005;11:82–86. Available from: https://www.ncbi.nlm.nih.gov/pubmed/15659950 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

