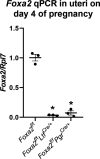
Targeted depletion of uterine glandular Foxa2 induces embryonic diapause in mice
- PMID: 35861728
- PMCID: PMC9355561
- DOI: 10.7554/eLife.78277
Targeted depletion of uterine glandular Foxa2 induces embryonic diapause in mice
Abstract
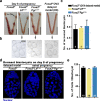
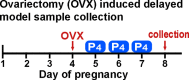
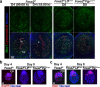
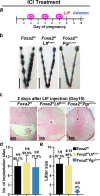
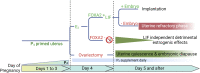
Embryonic diapause is a reproductive strategy in which embryo development and growth is temporarily arrested within the uterus to ensure the survival of neonates and mothers during unfavorable conditions. Pregnancy is reinitiated when conditions become favorable for neonatal survival. The mechanism of how the uterus enters diapause in various species remains unclear. Mice with uterine depletion of Foxa2, a transcription factor, are infertile. In this study, we show that dormant blastocysts are recovered from these mice on day 8 of pregnancy with persistent expression of uterine Msx1, a gene critical to maintaining the uterine quiescent state, suggesting that these mice enter embryonic diapause. Leukemia inhibitory factor (LIF) can resume implantation in these mice. Although estrogen is critical for implantation in progesterone-primed uterus, our current model reveals that FOXA2-independent estrogenic effects are detrimental to sustaining uterine quiescence. Interestingly, progesterone and anti-estrogen can prolong uterine quiescence in the absence of FOXA2. Although we find that Msx1 expression persists in the uterus deficient in Foxa2, the complex relationship of FOXA2 with Msx genes and estrogen receptors remains to be explored.
Keywords: Foxa2; delayed pregnancy; developmental biology; diapause; dormant; estrogen; mouse; uterus.
© 2022, Matsuo, Yuan, Kim et al.
Conflict of interest statement
MM, JY, YK, AD, HF, SD, XS No competing interests declared
Figures

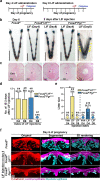
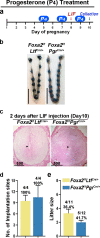
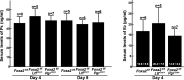
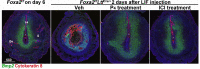
Similar articles
-
Foxa2-dependent uterine glandular cell differentiation is essential for successful implantation.Nat Commun. 2025 Mar 12;16(1):2465. doi: 10.1038/s41467-025-57848-w. Nat Commun. 2025. PMID: 40074766 Free PMC article.
-
Forkhead box a2 (FOXA2) is essential for uterine function and fertility.Proc Natl Acad Sci U S A. 2017 Feb 7;114(6):E1018-E1026. doi: 10.1073/pnas.1618433114. Epub 2017 Jan 3. Proc Natl Acad Sci U S A. 2017. PMID: 28049832 Free PMC article.
-
A new role for muscle segment homeobox genes in mammalian embryonic diapause.Open Biol. 2013 Apr 24;3(4):130035. doi: 10.1098/rsob.130035. Open Biol. 2013. PMID: 23615030 Free PMC article.
-
Embryonic and uterine factors in delayed implantation in rodents.J Reprod Fertil Suppl. 1981;29:159-72. J Reprod Fertil Suppl. 1981. PMID: 7014864 Review.
-
Mechanisms of uterine estrogen signaling during early pregnancy in mice: an update.J Mol Endocrinol. 2016 Apr;56(3):R127-38. doi: 10.1530/JME-15-0300. Epub 2016 Feb 17. J Mol Endocrinol. 2016. PMID: 26887389 Free PMC article. Review.
Cited by
-
mTOR activity paces human blastocyst stage developmental progression.Cell. 2024 Nov 14;187(23):6566-6583.e22. doi: 10.1016/j.cell.2024.08.048. Epub 2024 Sep 26. Cell. 2024. PMID: 39332412 Free PMC article.
-
Oxytocin induces embryonic diapause.Sci Adv. 2025 Mar 7;11(10):eadt1763. doi: 10.1126/sciadv.adt1763. Epub 2025 Mar 5. Sci Adv. 2025. PMID: 40043121 Free PMC article.
-
Murine uterine gland branching is necessary for gland function in implantation.Mol Hum Reprod. 2024 May 30;30(6):gaae020. doi: 10.1093/molehr/gaae020. Mol Hum Reprod. 2024. PMID: 38788747 Free PMC article.
-
Decoding Müllerian Duct Epithelial Regionalization.Mol Reprod Dev. 2025 Feb;92(2):e70018. doi: 10.1002/mrd.70018. Mol Reprod Dev. 2025. PMID: 39994938 Free PMC article. Review.
-
Foxa2-dependent uterine glandular cell differentiation is essential for successful implantation.Nat Commun. 2025 Mar 12;16(1):2465. doi: 10.1038/s41467-025-57848-w. Nat Commun. 2025. PMID: 40074766 Free PMC article.
References
-
- Cernilogar FM, Hasenöder S, Wang Z, Scheibner K, Burtscher I, Sterr M, Smialowski P, Groh S, Evenroed IM, Gilfillan GD, Lickert H, Schotta G. Pre-marked chromatin and transcription factor co-binding shape the pioneering activity of Foxa2. Nucleic Acids Research. 2019;47:9069–9086. doi: 10.1093/nar/gkz627. - DOI - PMC - PubMed
-
- Chen JR, Cheng JG, Shatzer T, Sewell L, Hernandez L, Stewart CL. Leukemia inhibitory factor can substitute for nidatory estrogen and is essential to inducing a receptive uterus for implantation but is not essential for subsequent embryogenesis. Endocrinology. 2000;141:4365–4372. doi: 10.1210/endo.141.12.7855. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

