Effect of NOTCH3 EGFr Group, Sex, and Cardiovascular Risk Factors on CADASIL Clinical and Neuroimaging Outcomes
- PMID: 35862191
- PMCID: PMC9508953
- DOI: 10.1161/STROKEAHA.122.039325
Effect of NOTCH3 EGFr Group, Sex, and Cardiovascular Risk Factors on CADASIL Clinical and Neuroimaging Outcomes
Abstract
Background: A retrospective study has shown that EGFr (epidermal growth factor-like repeat) group in the NOTCH3 gene is an important cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) disease modifier of age at first stroke and white matter hyperintensity (WMH) volume. No study has yet assessed the effect of other known CADASIL modifiers, that is, cardiovascular risk factors and sex, in the context of NOTCH3 EGFr group. In this study, we determined the relative disease-modifying effects of NOTCH3 EGFr group, sex and cardiovascular risk factor on disease severity in the first genotype-driven, large prospective CADASIL cohort study, using a comprehensive battery of CADASIL clinical outcomes and neuroimaging markers.
Methods: Patients with CADASIL participated in a single-center, prospective cohort study (DiViNAS [Disease Variability in NOTCH3 Associated Small Vessel Disease]) between 2017 and 2020. The study protocol included a clinical assessment, neuropsychological test battery and brain magnetic resonance imaging on a single research day. Multivariable linear, logistic and Cox regression models were used to cross-sectionally assess the effect of CADASIL modifiers on clinical severity (stroke, disability, processing speed) and neuroimaging markers (WMH volume, peak width of skeletonized mean diffusivity, lacune volume, brain volume, cerebral microbleed count).
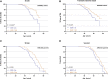
Results: Two hundred patients with CADASIL participated, of which 103 harbored a NOTCH3 EGFr 1-6 variant and 97 an EGFr 7-34 variant. NOTCH3 EGFr 1-6 group was the most important modifier of age at first stroke (hazard ratio, 2.45 [95% CI, 1.39-4.31]; P=0.002), lacune volume (odds ratio, 4.31 [95% CI, 2.31-8.04]; P=4.0×10-6), WMH volume (B=0.81 [95% CI, 0.60-1.02]; P=1.1×10-12), and peak width of skeletonized mean diffusivity (B=0.65 [95% CI, 0.44-0.87]; P=1.6×10-8). EGFr 1-6 patients had a significantly higher WMH volume in the anterior temporal lobes and superior frontal gyri and a higher burden of enlarged perivascular spaces. After NOTCH3 EGFr group, male sex and hypertension were the next most important modifiers of clinical outcomes and neuroimaging markers.
Conclusions: NOTCH3 EGFr group is the most important CADASIL disease modifier not only for age at first stroke and WMH volume but also strikingly so for a whole battery of clinically relevant disease measures such as lacune volume and peak width of skeletonized mean diffusivity. NOTCH3 EGFr group is followed in importance by sex, hypertension, diabetes, and smoking.
Keywords: CADASIL; hypertension; mutation; neuroimaging; risk factors; sex; white matter.
Figures




References
-
- Joutel A, Vahedi K, Corpechot C, Troesch A, Chabriat H, Vayssière C, Cruaud C, Maciazek J, Weissenbach J, Bousser MG, et al. . Strong clustering and stereotyped nature of Notch3 mutations in CADASIL patients. Lancet. 1997;350:1511–1515. doi: 10.1016/S0140-6736(97)08083-5 - PubMed
-
- Chabriat H, Joutel A, Dichgans M, Tournier-Lasserve E, Bousser MG. Cadasil. Lancet Neurol. 2009;8:643–653. doi: 10.1016/S1474-4422(09)70127-9 - PubMed
-
- Rutten JW, Hack RJ, Duering M, Gravesteijn G, Dauwerse JG, Overzier M, van den Akker EB, Slagboom E, Holstege H, Nho K, et al. . Broad phenotype of cysteine-altering NOTCH3 variants in UK Biobank: CADASIL to nonpenetrance. Neurology. 2020;95:e1835–e1843. doi: 10.1212/WNL.0000000000010525 - PMC - PubMed
-
- Rutten JW, Van Eijsden BJ, Duering M, Jouvent E, Opherk C, Pantoni L, Federico A, Dichgans M, Markus HS, Chabriat H, et al. . The effect of NOTCH3 pathogenic variant position on CADASIL disease severity: NOTCH3 EGFr 1-6 pathogenic variant are associated with a more severe phenotype and lower survival compared with EGFr 7-34 pathogenic variant. Genet Med. 2019;21:676–682. doi: 10.1038/s41436-018-0088-3 - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

