Discovery of Novel Iminosugar Compounds Produced by Lactobacillus paragasseri MJM60645 and Their Anti-Biofilm Activity against Streptococcus mutans
- PMID: 35863019
- PMCID: PMC9431463
- DOI: 10.1128/spectrum.01122-22
Discovery of Novel Iminosugar Compounds Produced by Lactobacillus paragasseri MJM60645 and Their Anti-Biofilm Activity against Streptococcus mutans
Abstract
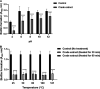
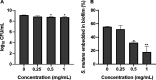
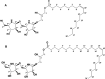
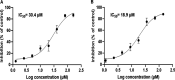
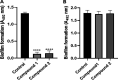
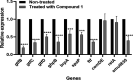
The oral cavity contains a number of microbes. They interact with each other and play an important role in human health. Among oral cariogenic microbes, Streptococcus mutans is recognized a major etiological bacteria of dental caries. Lactobacilli strains have been promoted as possible probiotic agents against S. mutans. However, their inhibitory mechanism has not been well elucidated yet. In the present study, two new compounds with strong antibiofilm activities were purified from the culture supernatant of Lactobacillus paragasseri MJM60645, which was isolated from the human oral cavity. These compounds showed strong inhibitory activities against S. mutans biofilm formation, with IC50 (concentration at which 50% biofilm was inhibited) of 30.4 μM for compound 1 and 18.9 μM for compound 2. However, these compounds did not show bactericidal activities against S. mutans. Structure elucidation by nuclear magnetic resonance (NMR) and mass spectrometry showed that compound 1 was composed of two arabinofuranose iminosugars jointed with one glycerol and oleic acid, and compound 2 was composed of two arabinofuranose iminosugars jointed with one glycerol and nervonic acid. To the best of our knowledge, these structures were discovered for the first time in this study. Treatment of S. mutans with compound 1 strongly downregulated expression levels of genes related to biofilm formation, including gtfB, gtfC, gtfD, gbpB, brpA, spaP, ftf, and smu0630 without affecting the expression of comDE or relA. This study provides new insights into novel molecules produced by Lactobacillus to regulate the pathogenesis of S. mutans, facilitating a better understanding of the mechanism for interactions between Lactobacillus and S. mutans. IMPORTANCE In this study, we isolated lactic acid bacteria that inhibit streptococcal biofilm from the oral cavity of infants and identified two novel compounds from the supernatant of their culture broth. The two compounds are structurally similar, and both consist of iminosugars, glycerol, and unsaturated fatty acid. A search of the SciFinder database revealed that these structures are novel and were discovered for the first time in this study. Mechanism studies have shown that these compounds can inhibit the expression of biofilm synthesis-related genes. This is the first report that lactic acid bacteria inhibit streptococcal biofilms by small molecules with new chemical structures. This study not only expands the understanding of natural products derived from lactic acid bacteria but also provides a new paradigm for the understanding of the interaction of bacteria in the oral microbiota.
Keywords: Lactobacillus paragasseri; Streptococcus mutans; biofilm; iminosugar; oral microbiota.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Inhibitory effect of Lactobacillus salivarius on Streptococcus mutans biofilm formation.Mol Oral Microbiol. 2015 Feb;30(1):16-26. doi: 10.1111/omi.12063. Epub 2014 Sep 8. Mol Oral Microbiol. 2015. PMID: 24961744
-
The enhancing antibiofilm activity of curcumin on Streptococcus mutans strains from severe early childhood caries.BMC Microbiol. 2020 Sep 16;20(1):286. doi: 10.1186/s12866-020-01975-5. BMC Microbiol. 2020. PMID: 32938379 Free PMC article.
-
Effect of Rubusoside, a Natural Sucrose Substitute, on Streptococcus mutans Biofilm Cariogenic Potential and Virulence Gene Expression In Vitro.Appl Environ Microbiol. 2020 Aug 3;86(16):e01012-20. doi: 10.1128/AEM.01012-20. Print 2020 Aug 3. Appl Environ Microbiol. 2020. PMID: 32503907 Free PMC article.
-
Antibacterial effects of natural compounds on biofilm formation of Streptococcus mutans.Clin Exp Dent Res. 2022 Dec;8(6):1426-1433. doi: 10.1002/cre2.673. Epub 2022 Oct 25. Clin Exp Dent Res. 2022. PMID: 36281582 Free PMC article. Review.
-
The virulence of Streptococcus mutans and the ability to form biofilms.Eur J Clin Microbiol Infect Dis. 2014 Apr;33(4):499-515. doi: 10.1007/s10096-013-1993-7. Epub 2013 Oct 24. Eur J Clin Microbiol Infect Dis. 2014. PMID: 24154653 Free PMC article. Review.
Cited by
-
Transcriptomic Analysis of Biofilm Formation Inhibition by PDIA Iminosugar in Staphylococcus aureus.Antibiotics (Basel). 2025 Jul 1;14(7):668. doi: 10.3390/antibiotics14070668. Antibiotics (Basel). 2025. PMID: 40723971 Free PMC article.
-
Small cyclic dipeptide produced by Lactobacillus rhamnosus with anti-biofilm properties against Streptococcus mutans biofilm.Biofilm. 2024 Nov 6;8:100237. doi: 10.1016/j.bioflm.2024.100237. eCollection 2024 Dec. Biofilm. 2024. PMID: 39624045 Free PMC article.
-
The Mechanisms and Application Value of Postbiotics in Caries Prevention and Management.Oral Health Prev Dent. 2024 Sep 12;22:465-478. doi: 10.3290/j.ohpd.b5740317. Oral Health Prev Dent. 2024. PMID: 39264370 Free PMC article. Review.
-
Anti-Quorum Sensing Activity of Probiotics: The Mechanism and Role in Food and Gut Health.Microorganisms. 2023 Mar 20;11(3):793. doi: 10.3390/microorganisms11030793. Microorganisms. 2023. PMID: 36985366 Free PMC article. Review.
-
Unveiling the dual nature of Lactobacillus: from cariogenic threat to probiotic protector-a critical review with bibliometric analysis.Front Oral Health. 2025 Jan 31;6:1535233. doi: 10.3389/froh.2025.1535233. eCollection 2025. Front Oral Health. 2025. PMID: 39959355 Free PMC article.
References
-
- Lamont RJ, Egland PG. 2015. Dental caries, p 945–955. In Tang YW, Sussman M, Liu D, Poxton I, Schwartzman J (ed), Molecular medical microbiology, 2nd ed. Academic Press, Boston, MA.
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

