Ontogeny of cellular organization and LGR5 expression in porcine cochlea revealed using tissue clearing and 3D imaging
- PMID: 35865132
- PMCID: PMC9294204
- DOI: 10.1016/j.isci.2022.104695
Ontogeny of cellular organization and LGR5 expression in porcine cochlea revealed using tissue clearing and 3D imaging
Abstract
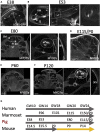
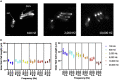
Over 11% of the world's population experience hearing loss. Although there are promising studies to restore hearing in rodent models, the size, ontogeny, genetics, and frequency range of hearing of most rodents' cochlea do not match that of humans. The porcine cochlea can bridge this gap as it shares many anatomical, physiological, and genetic similarities with its human counterpart. Here, we provide a detailed methodology to process and image the porcine cochlea in 3D using tissue clearing and light-sheet microscopy. The resulting 3D images can be employed to compare cochleae across different ages and conditions, investigate the ontogeny of cochlear cytoarchitecture, and produce quantitative expression maps of LGR5, a marker of cochlear progenitors in mice. These data reveal that hair cell organization, inner ear morphology, cellular cartography in the organ of Corti, and spatiotemporal expression of LGR5 are dynamic over developmental stages in a pattern not previously documented.
Keywords: Biological sciences research methodologies; Cell biology; Imaging anatomy; Optical imaging.
© 2022 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures






Similar articles
-
Three-dimensional imaging of intact porcine cochlea using tissue clearing and custom-built light-sheet microscopy.Biomed Opt Express. 2020 Oct 8;11(11):6181-6196. doi: 10.1364/BOE.402991. eCollection 2020 Nov 1. Biomed Opt Express. 2020. PMID: 33282483 Free PMC article.
-
Cellular cartography of the organ of Corti based on optical tissue clearing and machine learning.Elife. 2019 Jan 18;8:e40946. doi: 10.7554/eLife.40946. Elife. 2019. PMID: 30657453 Free PMC article.
-
Dynamic expression of Lgr5, a Wnt target gene, in the developing and mature mouse cochlea.J Assoc Res Otolaryngol. 2011 Aug;12(4):455-69. doi: 10.1007/s10162-011-0267-2. Epub 2011 Apr 7. J Assoc Res Otolaryngol. 2011. PMID: 21472479 Free PMC article.
-
An introduction to the genetics of normal and defective hearing.Ann N Y Acad Sci. 1997 Dec 29;830:361-74. doi: 10.1111/j.1749-6632.1997.tb51908.x. Ann N Y Acad Sci. 1997. PMID: 9616696 Review.
-
Expression and function of pannexins in the inner ear and hearing.BMC Cell Biol. 2016 May 24;17 Suppl 1(Suppl 1):16. doi: 10.1186/s12860-016-0095-7. BMC Cell Biol. 2016. PMID: 27229462 Free PMC article. Review.
Cited by
-
COMBINe enables automated detection and classification of neurons and astrocytes in tissue-cleared mouse brains.Cell Rep Methods. 2023 Apr 18;3(4):100454. doi: 10.1016/j.crmeth.2023.100454. eCollection 2023 Apr 24. Cell Rep Methods. 2023. PMID: 37159668 Free PMC article.
-
T-CLEARE: a pilot community-driven tissue clearing protocol repository.Front Bioeng Biotechnol. 2024 Sep 16;12:1304622. doi: 10.3389/fbioe.2024.1304622. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39351064 Free PMC article.
-
Surgical procedure of intratympanic injection and inner ear pharmacokinetics simulation in domestic pigs.Front Pharmacol. 2024 Jan 26;15:1348172. doi: 10.3389/fphar.2024.1348172. eCollection 2024. Front Pharmacol. 2024. PMID: 38344174 Free PMC article.
-
Tissue clearing and three-dimensional imaging of the whole cochlea and vestibular system from multiple large-animal models.STAR Protoc. 2023 Apr 13;4(2):102220. doi: 10.1016/j.xpro.2023.102220. Online ahead of print. STAR Protoc. 2023. PMID: 37060559 Free PMC article.
-
Spiner, deep learning-based automated detection of spiral ganglion neurons in intact cochleae.iScience. 2025 Jun 18;28(7):112929. doi: 10.1016/j.isci.2025.112929. eCollection 2025 Jul 18. iScience. 2025. PMID: 40687827 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Miscellaneous

