A conserved annexin A6-mediated membrane repair mechanism in muscle, heart, and nerve
- PMID: 35866481
- PMCID: PMC9431694
- DOI: 10.1172/jci.insight.158107
A conserved annexin A6-mediated membrane repair mechanism in muscle, heart, and nerve
Abstract
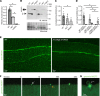
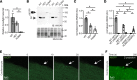
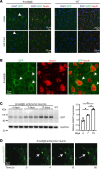
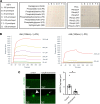
Membrane instability and disruption underlie myriad acute and chronic disorders. Anxa6 encodes the membrane-associated protein annexin A6 and was identified as a genetic modifier of muscle repair and muscular dystrophy. To evaluate annexin A6's role in membrane repair in vivo, we inserted sequences encoding green fluorescent protein (GFP) into the last coding exon of Anxa6. Heterozygous Anxa6gfp mice expressed a normal pattern of annexin A6 with reduced annexin A6GFP mRNA and protein. High-resolution imaging of wounded muscle fibers showed annexin A6GFP rapidly formed a repair cap at the site of injury. Injured cardiomyocytes and neurons also displayed repair caps after wounding, highlighting annexin A6-mediated repair caps as a feature in multiple cell types. Using surface plasmon resonance, we showed recombinant annexin A6 bound phosphatidylserine-containing lipids in a Ca2+- and dose-dependent fashion with appreciable binding at approximately 50 μM Ca2+. Exogenously added recombinant annexin A6 localized to repair caps and improved muscle membrane repair capacity in a dose-dependent fashion without disrupting endogenous annexin A6 localization, indicating annexin A6 promotes repair from both intracellular and extracellular compartments. Thus, annexin A6 orchestrates repair in multiple cell types, and recombinant annexin A6 may be useful in additional chronic disorders beyond skeletal muscle myopathies.
Keywords: Cardiology; Muscle; Muscle Biology; Neurological disorders.
Conflict of interest statement
Figures



Similar articles
-
Recombinant annexin A6 promotes membrane repair and protects against muscle injury.J Clin Invest. 2019 Nov 1;129(11):4657-4670. doi: 10.1172/JCI128840. J Clin Invest. 2019. PMID: 31545299 Free PMC article.
-
Annexin-A6 in Membrane Repair of Human Skeletal Muscle Cell: A Role in the Cap Subdomain.Cells. 2020 Jul 21;9(7):1742. doi: 10.3390/cells9071742. Cells. 2020. PMID: 32708200 Free PMC article.
-
Annexin A6 modifies muscular dystrophy by mediating sarcolemmal repair.Proc Natl Acad Sci U S A. 2014 Apr 22;111(16):6004-9. doi: 10.1073/pnas.1324242111. Epub 2014 Apr 9. Proc Natl Acad Sci U S A. 2014. PMID: 24717843 Free PMC article.
-
Annexins and Ca2+ handling in the heart.Cardiovasc Res. 2005 Mar 1;65(4):793-802. doi: 10.1016/j.cardiores.2004.11.010. Cardiovasc Res. 2005. PMID: 15721859 Review.
-
Annexin A6 in the liver: From the endocytic compartment to cellular physiology.Biochim Biophys Acta Mol Cell Res. 2017 Jun;1864(6):933-946. doi: 10.1016/j.bbamcr.2016.10.017. Epub 2016 Oct 27. Biochim Biophys Acta Mol Cell Res. 2017. PMID: 27984093 Review.
Cited by
-
Annexin A6 membrane repair protein protects against amyloid-induced dystrophic neurites and tau phosphorylation in Alzheimer's disease model mice.Acta Neuropathol. 2025 May 24;149(1):51. doi: 10.1007/s00401-025-02888-1. Acta Neuropathol. 2025. PMID: 40411591 Free PMC article.
-
AnnexinA6: a potential therapeutic target gene for extracellular matrix mineralization.Front Cell Dev Biol. 2023 Sep 4;11:1201200. doi: 10.3389/fcell.2023.1201200. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37727505 Free PMC article. Review.
-
Cells Responding to Closely Related Cholesterol-Dependent Cytolysins Release Extracellular Vesicles with a Common Proteomic Content Including Membrane Repair Proteins.Toxins (Basel). 2022 Dec 20;15(1):4. doi: 10.3390/toxins15010004. Toxins (Basel). 2022. PMID: 36668824 Free PMC article.
-
The extracellular matrix differentially directs myoblast motility and differentiation in distinct forms of muscular dystrophy: Dystrophic matrices alter myoblast motility.Matrix Biol. 2024 May;129:44-58. doi: 10.1016/j.matbio.2024.04.001. Epub 2024 Apr 4. Matrix Biol. 2024. PMID: 38582404 Free PMC article.
-
Physiological stress improves stem cell modeling of dystrophic cardiomyopathy.Dis Model Mech. 2024 Jun 1;17(6):dmm050487. doi: 10.1242/dmm.050487. Epub 2024 Feb 5. Dis Model Mech. 2024. PMID: 38050701 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

