Stop Codon Usage as a Window into Genome Evolution: Mutation, Selection, Biased Gene Conversion and the TAG Paradox
- PMID: 35867377
- PMCID: PMC9348620
- DOI: 10.1093/gbe/evac115
Stop Codon Usage as a Window into Genome Evolution: Mutation, Selection, Biased Gene Conversion and the TAG Paradox
Abstract
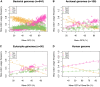
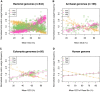
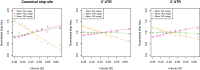
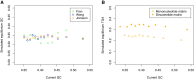
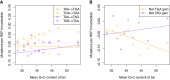
Protein coding genes terminate with one of three stop codons (TAA, TGA, or TAG) that, like synonymous codons, are not employed equally. With TGA and TAG having identical nucleotide content, analysis of their differential usage provides an unusual window into the forces operating on what are ostensibly functionally identical residues. Across genomes and between isochores within the human genome, TGA usage increases with G + C content but, with a common G + C → A + T mutation bias, this cannot be explained by mutation bias-drift equilibrium. Increased usage of TGA in G + C-rich genomes or genomic regions is also unlikely to reflect selection for the optimal stop codon, as TAA appears to be universally optimal, probably because it has the lowest read-through rate. Despite TAA being favored by selection and mutation bias, as with codon usage bias G + C pressure is the prime determinant of between-species TGA usage trends. In species with strong G + C-biased gene conversion (gBGC), such as mammals and birds, the high usage and conservation of TGA is best explained by an A + T → G + C repair bias. How to explain TGA enrichment in other G + C-rich genomes is less clear. Enigmatically, across bacterial and archaeal species and between human isochores TAG usage is mostly unresponsive to G + C pressure. This unresponsiveness we dub the TAG paradox as currently no mutational, selective, or gBGC model provides a well-supported explanation. That TAG does increase with G + C usage across eukaryotes makes the usage elsewhere yet more enigmatic. We suggest resolution of the TAG paradox may provide insights into either an unknown but common selective preference (probably at the DNA/RNA level) or an unrecognized complexity to the action of gBGC.
Keywords: genome evolution; molecular evolution; stop codon read-through; stop codon usage; translation termination; translational read-through.
© The Author(s) 2022. Published by Oxford University Press on behalf of Society for Molecular Biology and Evolution.
Figures

Similar articles
-
Unusual mammalian usage of TGA stop codons reveals that sequence conservation need not imply purifying selection.PLoS Biol. 2022 May 12;20(5):e3001588. doi: 10.1371/journal.pbio.3001588. eCollection 2022 May. PLoS Biol. 2022. PMID: 35550630 Free PMC article.
-
Stop codons in bacteria are not selectively equivalent.Biol Direct. 2012 Sep 13;7:30. doi: 10.1186/1745-6150-7-30. Biol Direct. 2012. PMID: 22974057 Free PMC article.
-
Variation in Release Factor Abundance Is Not Needed to Explain Trends in Bacterial Stop Codon Usage.Mol Biol Evol. 2022 Jan 7;39(1):msab326. doi: 10.1093/molbev/msab326. Mol Biol Evol. 2022. PMID: 34751397 Free PMC article.
-
Codon usage bias.Mol Biol Rep. 2022 Jan;49(1):539-565. doi: 10.1007/s11033-021-06749-4. Epub 2021 Nov 25. Mol Biol Rep. 2022. PMID: 34822069 Free PMC article. Review.
-
Codon usage and codon pair patterns in non-grass monocot genomes.Ann Bot. 2017 Nov 28;120(6):893-909. doi: 10.1093/aob/mcx112. Ann Bot. 2017. PMID: 29155926 Free PMC article. Review.
Cited by
-
Multiple independent genetic code reassignments of the UAG stop codon in phyllopharyngean ciliates.PLoS Genet. 2024 Dec 17;20(12):e1011512. doi: 10.1371/journal.pgen.1011512. eCollection 2024 Dec. PLoS Genet. 2024. PMID: 39689125 Free PMC article.
-
Lost in translation: conserved amino acid usage despite extreme codon bias in foraminifera.mBio. 2025 Apr 9;16(4):e0391624. doi: 10.1128/mbio.03916-24. Epub 2025 Mar 5. mBio. 2025. PMID: 40042280 Free PMC article.
-
Comparative genomics of two protozoans Dictyostelium discoideum and Plasmodium falciparum reveals conserved as well as distinct regulatory pathways crucial for exploring novel therapeutic targets for Malaria.Heliyon. 2024 Sep 26;10(19):e38500. doi: 10.1016/j.heliyon.2024.e38500. eCollection 2024 Oct 15. Heliyon. 2024. PMID: 39391471 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

