NeuroSAFE PROOF: study protocol for a single-blinded, IDEAL stage 3, multi-centre, randomised controlled trial of NeuroSAFE robotic-assisted radical prostatectomy versus standard robotic-assisted radical prostatectomy in men with localized prostate cancer
- PMID: 35869497
- PMCID: PMC9306247
- DOI: 10.1186/s13063-022-06421-7
NeuroSAFE PROOF: study protocol for a single-blinded, IDEAL stage 3, multi-centre, randomised controlled trial of NeuroSAFE robotic-assisted radical prostatectomy versus standard robotic-assisted radical prostatectomy in men with localized prostate cancer
Abstract
Background: Robotic radical prostatectomy (RARP) is a first-line curative treatment option for localized prostate cancer. Postoperative erectile dysfunction and urinary incontinence are common associated adverse side effects that can negatively impact patients' quality of life. Preserving the lateral neurovascular bundles (NS) during RARP improves functional outcomes. However, selecting men for NS may be difficult when there is concern about incurring in positive surgical margin (PSM) which in turn risks adverse oncological outcomes. The NeuroSAFE technique (intra-operative frozen section examination of the neurovascular structure adjacent prostate margin) can provide real-time pathological consult to promote optimal NS whilst avoiding PSM.
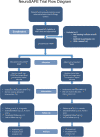
Methods: NeuroSAFE PROOF is a single-blinded, multi-centre, randomised controlled trial (RCT) in which men are randomly allocated 1:1 to either NeuroSAFE RARP or standard RARP. Men electing for RARP as primary treatment, who are continent and have good baseline erectile function (EF), defined by International Index of Erectile Function (IIEF-5) score > 21, are eligible. NS in the intervention arm is guided by the NeuroSAFE technique. NS in the standard arm is based on standard of care, i.e. a pre-operative image-based planning meeting, patient-specific clinical information, and digital rectal examination. The primary outcome is assessment of EF at 12 months. The primary endpoint is the proportion of men who achieve IIEF-5 score ≥ 21. A sample size of 404 was calculated to give a power of 90% to detect a difference of 14% between groups based on a feasibility study. Oncological outcomes are continuously monitored by an independent Data Monitoring Committee. Key secondary outcomes include urinary continence at 3 months assessed by the international consultation on incontinence questionnaire, rate of biochemical recurrence, EF recovery at 24 months, and difference in quality of life.
Discussion: NeuroSAFE PROOF is the first RCT of intra-operative frozen section during radical prostatectomy in the world. It is properly powered to evaluate a difference in the recovery of EF for men undergoing RARP assessed by patient-reported outcome measures. It will provide evidence to guide the use of the NeuroSAFE technique around the world.
Trial registration: NCT03317990 (23 October 2017). Regional Ethics Committee; reference 17/LO/1978.
Keywords: Frozen section; Nerve sparing; NeuroSAFE; Potency; Prostate cancer; Protocol; Randomised controlled trial; Robotic prostatectomy.
© 2022. The Author(s).
Conflict of interest statement
Within NeuroSAFE PROOF, laparoscopic ports are supplied by Applied Medical but they have had no role in the design, analysis, or collection of the data; in writing the manuscript; or in the decision to submit the manuscript for publication. The NeuroSAFE PROOF TMG reports no other interests to declare.
Figures


References
-
- Officen for National Statistics cancer registration data. https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/.... Accessed 18 May 2022.
-
- Hoffman KE, Penson DF, Zhao Z, Huang LC, Conwill R, Laviana AA, et al. Patient-reported outcomes through 5 years for Active surveillance, surgery, brachytherapy, or external beam radiation with or without androgen deprivation therapy for localized prostate cancer. JAMA. 2020;323(2):149–163. doi: 10.1001/jama.2019.20675. - DOI - PMC - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

