Development of a nanoparticle-based immunotherapy targeting PD-L1 and PLK1 for lung cancer treatment
- PMID: 35871223
- PMCID: PMC9308817
- DOI: 10.1038/s41467-022-31926-9
Development of a nanoparticle-based immunotherapy targeting PD-L1 and PLK1 for lung cancer treatment
Abstract
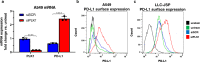
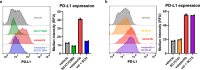
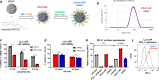
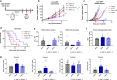
Immune checkpoint inhibitors (ICIs) targeting PD-L1 and PD-1 have improved survival in a subset of patients with advanced non-small cell lung cancer (NSCLC). However, only a minority of NSCLC patients respond to ICIs, highlighting the need for superior immunotherapy. Herein, we report on a nanoparticle-based immunotherapy termed ARAC (Antigen Release Agent and Checkpoint Inhibitor) designed to enhance the efficacy of PD-L1 inhibitor. ARAC is a nanoparticle co-delivering PLK1 inhibitor (volasertib) and PD-L1 antibody. PLK1 is a key mitotic kinase that is overexpressed in various cancers including NSCLC and drives cancer growth. Inhibition of PLK1 selectively kills cancer cells and upregulates PD-L1 expression in surviving cancer cells thereby providing opportunity for ARAC targeted delivery in a feedforward manner. ARAC reduces effective doses of volasertib and PD-L1 antibody by 5-fold in a metastatic lung tumor model (LLC-JSP) and the effect is mainly mediated by CD8+ T cells. ARAC also shows efficacy in another lung tumor model (KLN-205), which does not respond to CTLA-4 and PD-1 inhibitor combination. This study highlights a rational combination strategy to augment existing therapies by utilizing our nanoparticle platform that can load multiple cargo types at once.
© 2022. The Author(s).
Conflict of interest statement
OHSU, M.R., J.W.G., and W.Y. have a significant financial interest in PDX Pharmaceuticals, Inc., a company that may have a commercial interest in the results of this research and technology. This potential personal and institutional conflict of interest has been reviewed and managed by OHSU. The remaining authors declare no competing interests.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

