High-Yield Synthesis of Enantiopure 1,2-Amino Alcohols from l-Phenylalanine via Linear and Divergent Enzymatic Cascades
- PMID: 35873603
- PMCID: PMC9295148
- DOI: 10.1021/acs.oprd.1c00490
High-Yield Synthesis of Enantiopure 1,2-Amino Alcohols from l-Phenylalanine via Linear and Divergent Enzymatic Cascades
Abstract
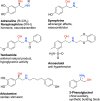
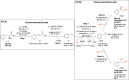
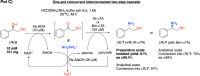
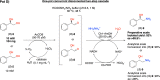
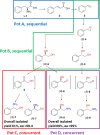
Enantiomerically pure 1,2-amino alcohols are important compounds due to their biological activities and wide applications in chemical synthesis. In this work, we present two multienzyme pathways for the conversion of l-phenylalanine into either 2-phenylglycinol or phenylethanolamine in the enantiomerically pure form. Both pathways start with the two-pot sequential four-step conversion of l-phenylalanine into styrene via subsequent deamination, decarboxylation, enantioselective epoxidation, and enantioselective hydrolysis. For instance, after optimization, the multienzyme process could convert 507 mg of l-phenylalanine into (R)-1-phenyl-1,2-diol in an overall isolated yield of 75% and >99% ee. The opposite enantiomer, (S)-1-phenyl-1,2-diol, was also obtained in a 70% yield and 98-99% ee following the same approach. At this stage, two divergent routes were developed to convert the chiral diols into either 2-phenylglycinol or phenylethanolamine. The former route consisted of a one-pot concurrent interconnected two-step cascade in which the diol intermediate was oxidized to 2-hydroxy-acetophenone by an alcohol dehydrogenase and then aminated by a transaminase to give enantiomerically pure 2-phenylglycinol. Notably, the addition of an alanine dehydrogenase enabled the connection of the two steps and made the overall process redox-self-sufficient. Thus, (S)-phenylglycinol was isolated in an 81% yield and >99.4% ee starting from ca. 100 mg of the diol intermediate. The second route consisted of a one-pot concurrent two-step cascade in which the oxidative and reductive steps were not interconnected. In this case, the diol intermediate was oxidized to either (S)- or (R)-2-hydroxy-2-phenylacetaldehyde by an alcohol oxidase and then aminated by an amine dehydrogenase to give the enantiomerically pure phenylethanolamine. The addition of a formate dehydrogenase and sodium formate was required to provide the reducing equivalents for the reductive amination step. Thus, (R)-phenylethanolamine was isolated in a 92% yield and >99.9% ee starting from ca. 100 mg of the diol intermediate. In summary, l-phenylalanine was converted into enantiomerically pure 2-phenylglycinol and phenylethanolamine in overall yields of 61% and 69%, respectively. This work exemplifies how linear and divergent enzyme cascades can enable the synthesis of high-value chiral molecules such as amino alcohols from a renewable material such as l-phenylalanine with high atom economy and improved sustainability.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
One-pot synthesis of (R)- and (S)-phenylglycinol from bio-based L-phenylalanine by an artificial biocatalytic cascade.Bioresour Bioprocess. 2021 Oct 6;8(1):97. doi: 10.1186/s40643-021-00448-5. Bioresour Bioprocess. 2021. PMID: 38650191 Free PMC article.
-
Microbial/enzymatic synthesis of chiral drug intermediates.Adv Appl Microbiol. 2000;47:33-78. doi: 10.1016/s0065-2164(00)47001-2. Adv Appl Microbiol. 2000. PMID: 12876794 Review.
-
One pot simultaneous preparation of both enantiomer of β-amino alcohol and vicinal diol via cascade biocatalysis.Biotechnol Lett. 2018 Feb;40(2):349-358. doi: 10.1007/s10529-017-2471-6. Epub 2017 Nov 9. Biotechnol Lett. 2018. PMID: 29124518
-
Stereo-Divergent Enzyme Cascades to Convert Racemic 4-Phenyl-2-Butanol into either (S)- or (R)-Corresponding Chiral Amine.Chembiochem. 2022 Apr 20;23(8):e202200108. doi: 10.1002/cbic.202200108. Epub 2022 Mar 3. Chembiochem. 2022. PMID: 35189014 Free PMC article.
-
Enzymatic synthesis of chiral intermediates for Omapatrilat, an antihypertensive drug.Biomol Eng. 2001 Jun;17(6):167-82. doi: 10.1016/s1389-0344(01)00068-5. Biomol Eng. 2001. PMID: 11337276 Review.
Cited by
-
Engineering Enzyme Substrate Scope Complementarity for Promiscuous Cascade Synthesis of 1,2-Amino Alcohols.Angew Chem Int Ed Engl. 2022 Nov 14;61(46):e202212637. doi: 10.1002/anie.202212637. Epub 2022 Oct 18. Angew Chem Int Ed Engl. 2022. PMID: 36136093 Free PMC article.
-
Direct Biocatalytic Processes for CO2 Capture as a Green Tool to Produce Value-Added Chemicals.Molecules. 2023 Jul 19;28(14):5520. doi: 10.3390/molecules28145520. Molecules. 2023. PMID: 37513391 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources
