Medial Septum Modulates Consciousness and Psychosis-Related Behaviors Through Hippocampal Gamma Activity
- PMID: 35874429
- PMCID: PMC9301478
- DOI: 10.3389/fncir.2022.895000
Medial Septum Modulates Consciousness and Psychosis-Related Behaviors Through Hippocampal Gamma Activity
Abstract
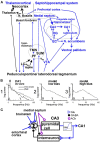
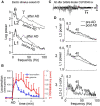
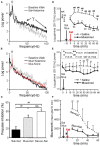
Abnormally high-amplitude hippocampal gamma activity (30-100 Hz) in behaving animals is seen after a hippocampal seizure, following injection of phencyclidine (PCP) or ketamine, and transiently in a delirium stage during induction of general anesthesia. High-amplitude hippocampal gamma activity in behaving rats is associated with hyperactive behavior and impairment in sensorimotor gating and sensory gating. The medial septum is necessary for the high-amplitude gamma activity and abnormal behaviors observed following a hippocampal seizure or injection of PCP/ketamine. Glutamatergic projection of the hippocampus to the nucleus accumbens (NAC) and dopaminergic transmission in NAC is necessary for abnormal behaviors. Large hippocampal gamma waves are suggested to contribute to seizure-induced automatism following temporal lobe seizures, and the schizophrenia-like symptoms induced by PCP/ketamine. Low-amplitude gamma activity is found during general anesthesia, associated with loss of consciousness in humans and loss of righting reflex in animals. Local inactivation or lesion of the medial septum, NAC, and brain areas connected to the septohippocampal-NAC system attenuates the increase in hippocampal gamma and associated behavioral disruptions induced by hippocampal seizure or PCP/ketamine. Inactivation or lesion of the septohippocampal-NAC system decreases the dose of anesthetic necessary for gamma decrease and loss of consciousness in animals. Thus, it is proposed that the septohippocampal-NAC system serves to control consciousness and the behavioral hyperactivity and neural dysfunctions during psychosis.
Keywords: gamma waves; general anesthesia; hippocampal seizure; ketamine; schizophrenia.
Copyright © 2022 Leung and Ma.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Adler L. E., Waldo M. C., Freedman R. (1985). Neurophysiologic studies of sensory gating in schizophrenia: comparison of auditory and visual responses. Biol. Psych. 20, 1284–1296. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

