Pegylated Liposomal Doxorubicin, Docetaxel, and Trastuzumab as Neoadjuvant Treatment for HER2-Positive Breast Cancer Patients: A Phase II and Biomarker Study
- PMID: 35875123
- PMCID: PMC9304895
- DOI: 10.3389/fonc.2022.909426
Pegylated Liposomal Doxorubicin, Docetaxel, and Trastuzumab as Neoadjuvant Treatment for HER2-Positive Breast Cancer Patients: A Phase II and Biomarker Study
Abstract
Background: Combined neoadjuvant chemotherapy with trastuzumab and pertuzumab is the standard regimen for human epidermal growth receptor 2 (HER2)-positive breast cancer (BC). However, pertuzumab is not available because it is not on the market or covered by medicare in some regions or poor economy. Anthracyclines and taxanes are cornerstones in BC chemotherapy, and their combination contributes to satisfactory efficiency in neoadjuvant settings. Nonetheless, concomitant administration of trastuzumab and an anthracycline is generally avoided clinically due to cardiotoxicity. Pegylated liposomal doxorubicin (PLD) is less cardiotoxic compared with traditional anthracyclines. Here, we conducted this prospective study to evaluate the efficacy, safety, and potential biomarkers for PLD plus trastuzumab and docetaxel as neoadjuvant treatment in HER2-positive BC.
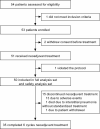
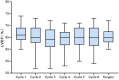
Patients and methods: Patients with stage II or III HER2-positive BC were recruited in this multicenter, open-label, single-arm, phase II study. Eligible patients were given 6 cycles of PLD plus docetaxel and trastuzumab. Primary endpoint was total pathological complete response (tpCR, ypT0/is ypN0). Secondary endpoints were breast pathological complete response (bpCR, ypT0/is), objective response rate (ORR), operation rate, breast-conserving surgery rate, and safety. Metadherin (MTDH), glutaminyl-peptide cyclotransferase (QPCT), topoisomerase II alpha (TOP2A), programmed death ligand 1 (PD-L1), and tumor-infiltrating lymphocytes (TILs) were evaluated in BC tissues pre-neoadjuvant for potential biomarkers.
Results: Between March 2019 and February 2021, 54 patients were enrolled, 50 were included in the analysis, and 35 (70.0%) completed 6 cycles of neoadjuvant treatment. Forty-nine (98.0%) patients underwent surgery with a breast-conserving rate of 44.0%. The tpCR rate, bpCR rate, and ORR were 48.0% (95% CI, 33.7%-62.6%), 60.0% (95% CI, 45.2%-73.6%), and 84.0% (95% CI, 70.9%-92.8%), respectively. tpCR was associated with MTDH (p = 0.002) and QPCT (p = 0.036) expression but not with TOP2A (p = 0.75), PD-L1 (p = 0.155), or TILs (p = 0.76). Patients with HR-negative status were more likely to achieve bpCR compared with those with HR-positive status (76.2% vs. 48.3%, p = 0.047). Grade ≥3 adverse events occurred in 38.0% of patients. Left ventricular ejection fraction decline by ≥10% was reported in 18.0% of patients, and no patient experienced congestive heart failure.
Conclusions: PLD plus docetaxel and trastuzumab might be a potential neoadjuvant regimen for HER2-positive BC with a high tpCR rate and manageable tolerability. MTDH and QPCT are potential predictive markers for tpCR.
Keywords: HER2-positive breast cancer; biomarker; efficacy; neoadjuvant treatment; pegylated liposomal doxorubicin; safety; trastuzumab.
Copyright © 2022 Wang, Li, Qi, Zhao, Kong, Yang, Yang, Zhang, Liu and Song.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Sequential neoadjuvant chemotherapy using pegylated liposomal doxorubicin and cyclophosphamide followed by taxanes with complete trastuzumab and pertuzumab treatment for HER2-positive breast cancer: A phase II single-arm study.Chin J Cancer Res. 2024 Feb 29;36(1):55-65. doi: 10.21147/j.issn.1000-9604.2024.01.06. Chin J Cancer Res. 2024. PMID: 38455369 Free PMC article.
-
Neoadjuvant trastuzumab, pertuzumab, and chemotherapy versus trastuzumab emtansine plus pertuzumab in patients with HER2-positive breast cancer (KRISTINE): a randomised, open-label, multicentre, phase 3 trial.Lancet Oncol. 2018 Jan;19(1):115-126. doi: 10.1016/S1470-2045(17)30716-7. Epub 2017 Nov 23. Lancet Oncol. 2018. PMID: 29175149 Clinical Trial.
-
Real-world effectiveness of dual HER2 blockade with pertuzumab and trastuzumab for neoadjuvant treatment of HER2-positive early breast cancer (The NEOPETRA Study).Breast Cancer Res Treat. 2020 Nov;184(2):469-479. doi: 10.1007/s10549-020-05866-1. Epub 2020 Sep 2. Breast Cancer Res Treat. 2020. PMID: 32876911
-
DECRESCENDO: de-escalating chemotherapy in HER2-positive, estrogen receptor-negative, node-negative early breast cancer.Future Oncol. 2023 Aug;19(24):1655-1667. doi: 10.2217/fon-2022-1282. Epub 2023 Aug 23. Future Oncol. 2023. PMID: 37609714 Review.
-
Risk-based decision-making in the treatment of HER2-positive early breast cancer: Recommendations based on the current state of knowledge.Cancer Treat Rev. 2021 Sep;99:102229. doi: 10.1016/j.ctrv.2021.102229. Epub 2021 May 20. Cancer Treat Rev. 2021. PMID: 34139476 Review.
Cited by
-
HER2-targeted therapies for HER2-positive early-stage breast cancer: present and future.Front Pharmacol. 2024 Sep 16;15:1446414. doi: 10.3389/fphar.2024.1446414. eCollection 2024. Front Pharmacol. 2024. PMID: 39351085 Free PMC article. Review.
-
Efficacy, toxicity and prognostic factors of pyrotinib‑involved neoadjuvant therapy in HER2‑positive breast cancer: A retrospective study.Oncol Lett. 2023 Jun 6;26(1):314. doi: 10.3892/ol.2023.13900. eCollection 2023 Jul. Oncol Lett. 2023. PMID: 37332338 Free PMC article.
-
LncRNA PTPRG-AS1 Promotes Breast Cancer Progression by Modulating the miR-4659a-3p/QPCT Axis.Onco Targets Ther. 2024 Oct 4;17:805-819. doi: 10.2147/OTT.S474898. eCollection 2024. Onco Targets Ther. 2024. PMID: 39380914 Free PMC article.
-
Imaging-proteomic analysis for prediction of neoadjuvant chemotherapy responses in patients with breast cancer.Cancer Med. 2023 Dec;12(23):21256-21269. doi: 10.1002/cam4.6704. Epub 2023 Nov 14. Cancer Med. 2023. PMID: 37962087 Free PMC article.
-
Pegylated liposomal doxorubicin + cyclophosphamide followed by taxane as adjuvant therapy for early-stage breast cancer: a randomized controlled trial.Oncologist. 2025 Jun 4;30(6):oyaf101. doi: 10.1093/oncolo/oyaf101. Oncologist. 2025. PMID: 40504550 Free PMC article. Clinical Trial.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

