Mussel-Inspired Polydopamine-Based Multilayered Coatings for Enhanced Bone Formation
- PMID: 35875492
- PMCID: PMC9301208
- DOI: 10.3389/fbioe.2022.952500
Mussel-Inspired Polydopamine-Based Multilayered Coatings for Enhanced Bone Formation
Abstract
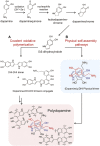
Repairing bone defects remains a challenge in clinical practice and the application of artificial scaffolds can enhance local bone formation, but the function of unmodified scaffolds is limited. Considering different application scenarios, the scaffolds should be multifunctionalized to meet specific demands. Inspired by the superior adhesive property of mussels, polydopamine (PDA) has attracted extensive attention due to its universal capacity to assemble on all biomaterials and promote further adsorption of multiple external components to form PDA-based multilayered coatings with multifunctional property, which can induce synergistic enhancement of new bone formation, such as immunomodulation, angiogenesis, antibiosis and antitumor property. This review will summarize mussel-inspired PDA-based multilayered coatings for enhanced bone formation, including formation mechanism and biofunction of PDA coating, as well as different functional components. The synergistic enhancement of multiple functions for better bone formation will also be discussed. This review will inspire the design and fabrication of PDA-based multilayered coatings for different application scenarios and promote deeper understanding of their effect on bone formation, but more efforts should be made to achieve clinical translation. On this basis, we present a critical conclusion, and forecast the prospects of PDA-based multilayered coatings for bone regeneration.
Keywords: bone regeneration; immunomodulation; multifunction; multilayered coating; osteogenesis; polydopamine.
Copyright © 2022 Wu, Zhao, Lin and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
[Advances in polydopamine surface modification for capillary electrochromatography].Se Pu. 2020 Sep 8;38(9):1057-1068. doi: 10.3724/SP.J.1123.2020.03004. Se Pu. 2020. PMID: 34213272 Chinese.
-
Improved in vitro angiogenic behavior of human umbilical vein endothelial cells with oxidized polydopamine coating.Colloids Surf B Biointerfaces. 2020 Oct;194:111176. doi: 10.1016/j.colsurfb.2020.111176. Epub 2020 Jun 6. Colloids Surf B Biointerfaces. 2020. PMID: 32540767
-
Mussel-inspired polydopamine-mediated surface modification of freeze-cast poly (ε-caprolactone) scaffolds for bone tissue engineering applications.Biomed Tech (Berl). 2020 May 26;65(3):273-287. doi: 10.1515/bmt-2019-0061. Biomed Tech (Berl). 2020. PMID: 31655791
-
Integration of inorganic nanostructures with polydopamine-derived carbon: tunable morphologies and versatile applications.Nanoscale. 2016 Jan 28;8(4):1770-88. doi: 10.1039/c5nr06711a. Nanoscale. 2016. PMID: 26750427 Review.
-
Recent Development and Applications of Polydopamine in Tissue Repair and Regeneration Biomaterials.Int J Nanomedicine. 2024 Jan 25;19:859-881. doi: 10.2147/IJN.S437854. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38293610 Free PMC article. Review.
Cited by
-
New liquid supports in the development of integrated platforms for the reuse of oxidative enzymes and polydopamine production.Front Bioeng Biotechnol. 2022 Nov 28;10:1037322. doi: 10.3389/fbioe.2022.1037322. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36518198 Free PMC article.
-
Simultaneous detection of neurotransmitters and Cu2+ using double-bore carbon fiber microelectrodes via fast-scan cyclic voltammetry.RSC Adv. 2023 Nov 20;13(48):33844-33851. doi: 10.1039/d3ra06218j. eCollection 2023 Nov 16. RSC Adv. 2023. PMID: 38020012 Free PMC article.
-
Silk fibroin scaffolds: A promising candidate for bone regeneration.Front Bioeng Biotechnol. 2022 Nov 25;10:1054379. doi: 10.3389/fbioe.2022.1054379. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36507269 Free PMC article. Review.
-
Mussel-Based Biomimetic Strategies in Musculoskeletal Disorder Treatment: From Synthesis Principles to Diverse Applications.Int J Nanomedicine. 2023 Jan 24;18:455-472. doi: 10.2147/IJN.S386635. eCollection 2023. Int J Nanomedicine. 2023. PMID: 36718191 Free PMC article. Review.
-
Exploring the biological activity and setting dynamics of a novel polydopamine-based root repair material.J Oral Biol Craniofac Res. 2025 Jul-Aug;15(4):737-743. doi: 10.1016/j.jobcr.2025.04.001. Epub 2025 Apr 29. J Oral Biol Craniofac Res. 2025. PMID: 40352478 Free PMC article.
References
-
- Albashari A. A., He Y., Albaadani M. A., Xiang Y., Ali J., Hu F., et al. (2021). Titanium Nanotube Modified with Silver Cross-Linked Basic Fibroblast Growth Factor Improves Osteoblastic Activities of Dental Pulp Stem Cells and Antibacterial Effect. Front. Cell. Dev. Biol. 9, 654654. 10.3389/fcell.2021.654654 - DOI - PMC - PubMed
-
- Asadi H., Ghalei S., Handa H., Ramasamy R. P. (2021). Cellulose Nanocrystal Reinforced Silk Fibroin Coating for Enhanced Corrosion Protection and Biocompatibility of Mg-Based Alloys for Orthopedic Implant Applications. Prog. Org. Coatings 161, 106525. 10.1016/j.porgcoat.2021.106525 - DOI
Publication types
LinkOut - more resources
Full Text Sources

