Sonicated Bordetella bronchiseptica Bacterin Can Protect Dendritic Cells from Differential Cytotoxicity Caused by Doxorubicin and Vincristine and Enhance Their Antigen-Presenting Capability
- PMID: 35877437
- PMCID: PMC9323433
- DOI: 10.3390/cimb44070213
Sonicated Bordetella bronchiseptica Bacterin Can Protect Dendritic Cells from Differential Cytotoxicity Caused by Doxorubicin and Vincristine and Enhance Their Antigen-Presenting Capability
Abstract
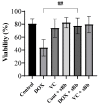
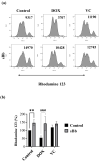
Doxorubicin (DOX) and vincristine (VC) are anti-cancer drugs commonly used for lymphoma in veterinary and human medicine. However, there are several side effects caused by these drugs. In this study, the protective effects of sonicated Bordetella bronchiseptica bacterin (sBb) on dendritic cells (DCs) damaged by two anti-cancer drugs were investigated. DCs play important roles in the innate and adaptive immunity of hosts, especially activating T cells that can suppress tumor growth. The metabolic activity of DCs significantly increased after the treatment with sBb compared to that of control DCs. In addition, there was a marked change in mitochondrial integrity between DOX-treated DC and DOX + sBb-treated DCs. Flow cytometric analysis also demonstrated that sBb upregulated the expression of the surface markers of DCs, particularly CD54. In mixed lymphocyte responses, sBb significantly increased the antigen-presenting capability of DCs. In particular, sBb increased the capability of control DCs by approximately 150% and that of VC-treated DCs by 221%. These results suggest that sBb can be used as a potential immunostimulatory agent to protect DCs from anti-cancer drug-induced damage and provide fundamental information about using a combination of DCs and vincristine in immunotherapy.
Keywords: dendritic cell; doxorubicin; protective effects; sonicated Bordetella bronchiseptica; vincristine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Immunostimulatory activity and safety evaluation of Bordetella bronchiseptica-derived lipopolysaccharide, a new vaccine adjuvant candidate.Immunobiology. 2023 Sep;228(5):152709. doi: 10.1016/j.imbio.2023.152709. Epub 2023 Jul 17. Immunobiology. 2023. PMID: 37487385
-
Bordetella bronchiseptica is a potent and safe adjuvant that enhances the antigen-presenting capability of dendritic cells.Korean J Physiol Pharmacol. 2020 Jan;24(1):47-52. doi: 10.4196/kjpp.2020.24.1.47. Epub 2020 Dec 20. Korean J Physiol Pharmacol. 2020. PMID: 31908574 Free PMC article.
-
Bordetella type III secretion and adenylate cyclase toxin synergize to drive dendritic cells into a semimature state.J Immunol. 2004 Aug 1;173(3):1934-40. doi: 10.4049/jimmunol.173.3.1934. J Immunol. 2004. PMID: 15265927
-
Genetically modified dendritic cells in cancer therapy: implications for transfusion medicine.Transfus Med Rev. 2001 Oct;15(4):292-304. doi: 10.1053/tmrv.2001.26960. Transfus Med Rev. 2001. PMID: 11668436 Review.
-
Dendritic Cells in Oncolytic Virus-Based Anti-Cancer Therapy.Viruses. 2015 Dec 9;7(12):6506-25. doi: 10.3390/v7122953. Viruses. 2015. PMID: 26690204 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

