An Engineered Protein-Based Building Block (Albumin Methacryloyl) for Fabrication of a 3D In Vitro Cryogel Model
- PMID: 35877489
- PMCID: PMC9324498
- DOI: 10.3390/gels8070404
An Engineered Protein-Based Building Block (Albumin Methacryloyl) for Fabrication of a 3D In Vitro Cryogel Model
Abstract
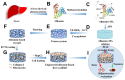
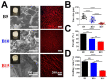
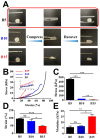
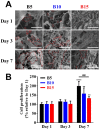
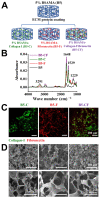
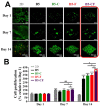
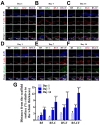
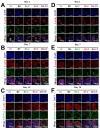
Drug-induced liver injury (DILI) is a leading cause of attrition in drug development or withdrawal; current animal experiments and traditional 2D cell culture systems fail to precisely predict the liver toxicity of drug candidates. Hence, there is an urgent need for an alternative in vitro model that can mimic the liver microenvironments and accurately detect human-specific drug hepatotoxicity. Here, for the first time we propose the fabrication of an albumin methacryloyl cryogel platform inspired by the liver's microarchitecture via emulating the mechanical properties and extracellular matrix (ECM) cues of liver. Engineered crosslinkable albumin methacryloyl is used as a protein-based building block for fabrication of albumin cryogel in vitro models that can have potential applications in 3D cell culture and drug screening. In this work, protein modification, cryogelation, and liver ECM coating were employed to engineer highly porous three-dimensional cryogels with high interconnectivity, liver-like stiffness, and liver ECM as artificial liver constructs. The resulting albumin-based cryogel in vitro model provided improved cell-cell and cell-material interactions and consequently displayed excellent liver functional gene expression, being conducive to detection of fialuridine (FIAU) hepatotoxicity.
Keywords: 3D in vitro models; albumin methacryloyl; cryogels; liver tissue engineering.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources

