Varenicline for Gradual Versus Abrupt Smoking Cessation in Poorly Motivated Smokers With COPD: A Prematurely Terminated Randomized Controlled Trial
- PMID: 35877930
- PMCID: PMC9718580
- DOI: 10.15326/jcopdf.2022.0305
Varenicline for Gradual Versus Abrupt Smoking Cessation in Poorly Motivated Smokers With COPD: A Prematurely Terminated Randomized Controlled Trial
Abstract
Background: Although smoking is the leading cause of chronic obstructive pulmonary disease (COPD), many patients with COPD smoke, highlighting the need for effective smoking cessation interventions in this population. This study examined the efficacy and safety of varenicline in increasing smoking cessation rates through "gradual" versus "abrupt" cessation in COPD patients with low motivation to quit smoking.
Methods: A randomized, open label, 30-week, controlled trial (ClinicalTrials.gov identifier: NCT02894957) was conducted between January 2019 and October 2020 at a center in Israel. Smokers with COPD, poorly motivated to quit, were randomized to 6 weeks of varenicline for smoking reduction and a target quit day (TQD) at the end of week 6 (gradual cessation group) or ad libitum smoking for 5 weeks, 1 week of varenicline, and a TQD at the end of week 6 (abrupt cessation group). After the pre-quit phase, both groups received 12-week regular varenicline treatment and 12-week follow-up. Primary outcome was biochemically-validated continuous abstinence for weeks 6-30. Secondary outcomes were: (1) biochemically-confirmed7-day point prevalence abstinence for weeks 4-30, (2) efficient smoking reduction (≥50% in number of cigarettes/day) in the pre-quit phase; and (3) number of cigarettes/day, motivation to quit, and changes in respiratory symptoms and spirometry from baseline through week 30.
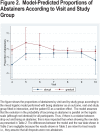
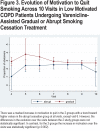
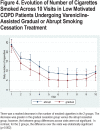
Results: A drug recall issued by the study sponsor stopped the study after 70/242 (28.9%) patients had been enrolled. The gradual cessation group (n=29) had significantly higher continuous abstinence rates from TQD through week 30 versus the abrupt cessation group (n=41): 20.7% versus 4.9% (odds ratio [OR]=5.09; 95% confidence interval [CI] 0.89-29.17; p=0.048) and higher 7-day point prevalence abstinence levels at all time points but week 18 (p=0.027 at week 6, 0.056 at week 7, and 0.096 at week 9). Motivation to quit increased (p=0.002) and the number of cigarettes/day decreased (p=0.002) over time in both groups. Respiratory symptoms, but not spirometry, improved in both groups at week 30. Treatment was safe and well tolerated.
Conclusion: In poorly motivated smokers with COPD, using varenicline for a 6-week gradual smoking cessation before TQD, compared with abrupt cessation, significantly increased quit rates up to 6 months. Results were not affected by the smaller-than-expected sample size. Further studies are needed to confirm these data.
Keywords: chronic obstructive pulmonary disease; cigarette smoking; copd; smoking; smoking cessation; varenicline.
JCOPDF © 2022.
Conflict of interest statement
The authors have no interests to declare.
References
-
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease, 2022 report. Updated November 2021. Accessed January 2022. http://www.goldcopd.org
-
- Lundback B,Lindbeg A,Lindstrom M,et al. Not 15 but 50% of smokers develop COPD? Report from the obstructive lung disease in northern Sweden studies. Respir Med. 2003;97:115-122. doi: https://doi.org/10.1053/rmed.2003.1446 - PubMed
-
- Anthonisen NR,Connett JE,Kiley JP,et al. Effects of smoking intervention and the use of an inhaled anticholinergic bronchodilator on the rate of decline of FEV1. The lung health study. JAMA. 1994;272(19):1497-1505. doi: https://doi.org/10.1001/jama.1994.03520190043033 - PubMed
-
- Anthonisen NA,Connet JE,Murray RP. Smoking and lung function of lung health study participants after 11 years. Am J Respir Crit Care Med. 2002;166(5):675-679. doi: https://doi.org/10.1164/rccm.2112096 - PubMed
-
- Schauer GL,Wheaton AG,Malarcher AM,Croft JB. Smoking prevalence and cessation characteristics among U.S. adults with and without COPD: findings from the 2011 behavioral risk factor surveillance system. COPD. 2014;11(6):697-704. doi: https://doi.org/10.3109/15412555.2014.898049 - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical