Cytoprotective effects and antioxidant activities of acteoside and various extracts of Clerodendrum cyrtophyllum Turcz leaves against t-BHP induced oxidative damage
- PMID: 35879416
- PMCID: PMC9314432
- DOI: 10.1038/s41598-022-17038-w
Cytoprotective effects and antioxidant activities of acteoside and various extracts of Clerodendrum cyrtophyllum Turcz leaves against t-BHP induced oxidative damage
Abstract
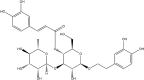
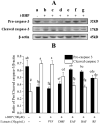
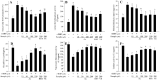
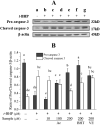
This study evaluates the antioxidant potential and cytoprotective effects of ethanolic crude extract from Clerodendrum cyrtophyllum leaves (ECE) and five derived fractions (namely, petroleum ether fraction (PEF), dichloromethane fraction (DMF), ethyl acetate fraction (EAF), n-butyl alcohol fraction (BAF) and the remaining fraction (RF)), as well as acteoside (Ac, a major phenolic component in EAF) on oxidative damage caused by tert-butyl hydroperoxide (t-BHP) in HepG2 cells. MTT assay results showed that ECE, EAF, BAF, RF and Ac increased the viability of t-BHP-damaged cells in a dose-dependent manner, while EAF significantly promoted cell viability. EAF, BAF, RF, or Ac reduced the levels of lactate dehydrogenase (LDH) leakage, malondialdehyde (MDA), and reactive oxygen species (ROS). Additionally, glutathione (GSH) levels and the activities of superoxide dismutase (SOD) and catalase (CAT) increased. Western blot analysis further indicated that EAF, BAF, RF, or Ac up-regulated pro-caspase-3 and reduced cleaved caspase-3 during t-BHP-induced oxidative stress. Flow cytometry analysis and fluorescence micrographs showed that Ac could inhibit apoptosis.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Antioxidant activities of Clerodendrum cyrtophyllum Turcz leaf extracts and their major components.PLoS One. 2020 Jun 23;15(6):e0234435. doi: 10.1371/journal.pone.0234435. eCollection 2020. PLoS One. 2020. PMID: 32574221 Free PMC article.
-
Ethyl Acetate Fraction of Hemerocallis citrina Baroni Decreases Tert-butyl Hydroperoxide-Induced Oxidative Stress Damage in BRL-3A Cells.Oxid Med Cell Longev. 2018 Nov 8;2018:1526125. doi: 10.1155/2018/1526125. eCollection 2018. Oxid Med Cell Longev. 2018. PMID: 30538798 Free PMC article.
-
Phytochemical characterization and hepatoprotective effect of active fragment from Adhatoda vasica Nees. against tert-butyl hydroperoxide induced oxidative impairment via activating AMPK/p62/Nrf2 pathway.J Ethnopharmacol. 2021 Feb 10;266:113454. doi: 10.1016/j.jep.2020.113454. Epub 2020 Oct 13. J Ethnopharmacol. 2021. PMID: 33065254
-
Protective effects of germinated and fermented soybean extract against tert-butyl hydroperoxide-induced hepatotoxicity in HepG2 cells and in rats.Food Funct. 2015 Nov;6(11):3512-21. doi: 10.1039/c5fo00785b. Epub 2015 Aug 24. Food Funct. 2015. PMID: 26299642
-
Meroterpenoid-Rich Fraction of the Ethanolic Extract from Sargassum serratifolium Suppressed Oxidative Stress Induced by Tert-Butyl Hydroperoxide in HepG2 Cells.Mar Drugs. 2018 Oct 9;16(10):374. doi: 10.3390/md16100374. Mar Drugs. 2018. PMID: 30304831 Free PMC article.
Cited by
-
Multifaceted Biological Properties of Verbascoside/Acteoside: Antimicrobial, Cytotoxic, Anti-Inflammatory, and Immunomodulatory Effects.Antibiotics (Basel). 2025 Jul 11;14(7):697. doi: 10.3390/antibiotics14070697. Antibiotics (Basel). 2025. PMID: 40724000 Free PMC article. Review.
-
Inorganic Nitrogen-Containing Aerosol Deposition Caused "Excessive Photosynthesis" of Herbs, Resulting in Increased Nitrogen Demand.Plants (Basel). 2022 Aug 27;11(17):2225. doi: 10.3390/plants11172225. Plants (Basel). 2022. PMID: 36079607 Free PMC article.
-
Oligomeric Proanthocyanidins: An Updated Review of Their Natural Sources, Synthesis, and Potentials.Antioxidants (Basel). 2023 Apr 26;12(5):1004. doi: 10.3390/antiox12051004. Antioxidants (Basel). 2023. PMID: 37237870 Free PMC article. Review.
References
-
- Wang B, et al. Isolation and characterisation of five novel antioxidant peptides from ethanol-soluble proteins hydrolysate of spotless smoothhound (Mustelus griseus) muscle. J. Funct. Foods. 2014;6:176–185. doi: 10.1016/j.jff.2013.10.004. - DOI
-
- Nimse SB, Pal D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015;5:27986–28006. doi: 10.1039/c4ra13315c. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

