Insulin Glargine 300 U/mL Therapy in Children and Adolescents with Type 1 Diabetes
- PMID: 35881330
- PMCID: PMC9439977
- DOI: 10.1007/s40272-022-00520-3
Insulin Glargine 300 U/mL Therapy in Children and Adolescents with Type 1 Diabetes
Abstract
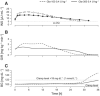
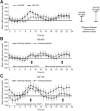
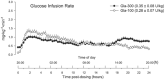
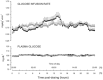
The pharmacokinetic and pharmacodynamic properties of the second-generation basal insulin glargine 300 Units/mL (Gla-300) may be of benefit in the treatment of type 1 diabetes mellitus (T1DM). Gla-300 provides a stable and sustained time-action profile, which is associated with glycaemic control and flexible dosing schedule. This review summarises the available evidence on the safety and efficacy of Gla-300 in children and adolescents with T1DM. Gla-300 is as effective as the first-generation basal insulin glargine 100 Units/mL (Gla-100), a standard of care for patients with diabetes in reducing HbA1c, and shows a lower risk of severe hypoglycaemia and hyperglycaemia in children and adolescents with T1DM. However, Gla-300 and Gla-100 are not bioequivalent and are not directly interchangeable. Real-world studies on patients aged 6-17 years are limited. To date, only one small study assessed the effectiveness and safety of Gla-300 versus Gla-100 in newly diagnosed T1DM paediatric patients, confirming the treatment safety and effectiveness of Gla-300 in clinical practice. Gla-300 is a longer-acting basal insulin alternative in the management of children (aged ≥ 6 years) and adolescents with T1DM.
Plain language summary
The smooth and prolonged activity of insulin glargine 300 Units/mL (Gla-300), a second-generation basal insulin, results in a stable and sustained glycaemic control while allowing flexible dosing times. In children aged ≥ 6 years and adolescents with type 1 diabetes mellitus (T1DM), Gla-300 provides a glycaemic control comparable to that of glargine 100 Units/mL (Gla-100), a standard of care in patients with T1DM. Gla-300 and Gla-100 show similar safety profiles, with Gla-300 resulting in a clinically relevant trend towards a lower incidence of hyperglycaemia with ketosis and a lower incidence and event rate of severe hypoglycaemia in children and adolescents with T1DM. Overall, these two insulins are similar, but not interchangeable. To the best of our knowledge, only one real-world study has addressed the effectiveness and safety of Gla-300 compared to Gla-100 in newly diagnosed T1DM paediatric patients, and it has shown the therapeutic benefits of Gla-300 in clinical practice.
© 2022. The Author(s).
Conflict of interest statement
Claudio Maffeis has received honoraria from Sanofi, Eli Lilly, Novo Nordisk and Sandoz for lectures including service on speaker bureaus and consulting fees from Abbot, Roche and Sanofi. Ivana Rabbone has received honoraria from Sanofi, Eli Lilly, Abbott, Aboca, Theras and Ypsomed for lectures including service on speaker bureaus.
Figures

Similar articles
-
Insulin Glargine in Type 1 Diabetes Mellitus: A Review of Clinical Trials and Real-world Evidence Across Two Decades.Curr Diabetes Rev. 2024;20(1):e100323214554. doi: 10.2174/1573399819666230310150905. Curr Diabetes Rev. 2024. PMID: 36896906 Free PMC article. Review.
-
Glycaemic control and hypoglycaemia during 12 months of randomized treatment with insulin glargine 300 U/mL versus glargine 100 U/mL in people with type 1 diabetes (EDITION 4).Diabetes Obes Metab. 2018 Jan;20(1):121-128. doi: 10.1111/dom.13048. Epub 2017 Aug 8. Diabetes Obes Metab. 2018. PMID: 28661585 Free PMC article. Clinical Trial.
-
Lower risk of severe hypoglycaemia with insulin glargine 300 U/mL versus glargine 100 U/mL in participants with type 1 diabetes: A meta-analysis of 6-month phase 3 clinical trials.Diabetes Obes Metab. 2020 Oct;22(10):1880-1885. doi: 10.1111/dom.14109. Epub 2020 Jul 21. Diabetes Obes Metab. 2020. PMID: 32515543 Free PMC article.
-
A review of the safety and efficacy data for insulin glargine 300 units/ml, a new formulation of insulin glargine.Diabetes Obes Metab. 2015 Dec;17(12):1107-14. doi: 10.1111/dom.12531. Epub 2015 Sep 10. Diabetes Obes Metab. 2015. PMID: 26139151 Review.
-
Efficacy and Safety of Insulin Glargine 300 Units/mL (Gla-300) Versus Insulin Glargine 100 Units/mL (Gla-100) in Children and Adolescents (6-17 years) With Type 1 Diabetes: Results of the EDITION JUNIOR Randomized Controlled Trial.Diabetes Care. 2020 Jul;43(7):1512-1519. doi: 10.2337/dc19-1926. Epub 2020 May 19. Diabetes Care. 2020. PMID: 32430458 Free PMC article. Clinical Trial.
Cited by
-
Effectiveness of switching from first-generation basal insulin to Glargine 300 U/mL in children and adolescents with type 1 diabetes: results from the ISPED CARD database.Acta Diabetol. 2024 Sep;61(9):1169-1176. doi: 10.1007/s00592-024-02304-2. Epub 2024 May 24. Acta Diabetol. 2024. PMID: 38789610
References
-
- Diabetes C, Complications Trial Research G. Nathan DM, Genuth S, Lachin J, Cleary P, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977–986. doi: 10.1056/NEJM199309303291401. - DOI - PubMed
-
- Control D, Groupa CTR Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: Diabetes Control and Complications Trial. J Pediatr. 1994;125(2):177–188. doi: 10.1016/S0022-3476(94)70190-3. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

