Enteropathogenic Escherichia coli (EPEC) expressing a non-functional bundle-forming pili (BFP) also leads to increased growth failure and intestinal inflammation in C57BL/6 mice
- PMID: 35882715
- PMCID: PMC9679052
- DOI: 10.1007/s42770-022-00802-5
Enteropathogenic Escherichia coli (EPEC) expressing a non-functional bundle-forming pili (BFP) also leads to increased growth failure and intestinal inflammation in C57BL/6 mice
Abstract
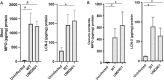
Bundle-forming pili (BFP) are implicated in the virulence of typical enteropathogenic E. coli (EPEC), resulting in enhanced colonization and mild to severe disease outcomes; hence, non-functional BFP may have a major influence on disease outcomes in vivo. Weaned antibiotic pre-treated C57BL/6 mice were orally infected with EPEC strain UMD901 (E2348/69 bfpA C129S); mice were monitored daily for body weight; stool specimens were collected daily; and intestinal tissues were collected at the termination of the experiment on day 3 post-infection. Real-time PCR was used to quantify fecal shedding and tissue burden. Intestinal inflammatory biomarkers lipocalin-2 (LCN-2) and myeloperoxidase (MPO) were also assessed. Infection caused substantial body weight loss, bloody diarrhea, and intestinal colonization with fecal and intestinal tissue inflammatory biomarkers that were comparable to those previously published with the wild-type typical EPEC strain. Here we further report on the evaluation of an EPEC infection model, showing how disruption of bfp function does not impair, and may even worsen diarrhea, colonization, and intestinal disruption and inflammation. More research is needed to understand the role of bfp in pathogenicity of EPEC infections in vivo.
Keywords: Bundle-forming pili; Diarrhea; Enteropathogenic E. coli; Inflammation; Murine model.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- Platts-Mills JA, Liu J, Rogawski ET, Kabir F, Lertsethtakarn P, Siguas M, Khan SS, Praharaj I, Murei A, Nshama R, et al. Use of quantitative molecular diagnostic methods to assess the aetiology, burden, and clinical characteristics of diarrhoea in children in low-resource settings: a reanalysis of the MAL-ED cohort study. Lancet Glob Health. 2018;6(12):e1309–e1318. doi: 10.1016/S2214-109X(18)30349-8. - DOI - PMC - PubMed
-
- Kotloff KL, Nataro JP, Blackwelder WC, Nasrin D, Farag TH, Panchalingam S, Wu Y, Sow SO, Sur D, Breiman RF, et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): a prospective, case-control study. Lancet. 2013;382(9888):209–222. doi: 10.1016/S0140-6736(13)60844-2. - DOI - PubMed
-
- Mundy R, Girard F, FitzGerald AJ, Frankel G. Comparison of colonization dynamics and pathology of mice infected with enteropathogenic Escherichia coli, enterohaemorrhagic E. coli and Citrobacter rodentium. FEMS Microbiol Lett. 2006;265(1):126–132. doi: 10.1111/j.1574-6968.2006.00481.x. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

