Increase in Anticancer Drug-Induced Toxicity by Fisetin in Lung Adenocarcinoma A549 Spheroid Cells Mediated by the Reduction of Claudin-2 Expression
- PMID: 35886884
- PMCID: PMC9316057
- DOI: 10.3390/ijms23147536
Increase in Anticancer Drug-Induced Toxicity by Fisetin in Lung Adenocarcinoma A549 Spheroid Cells Mediated by the Reduction of Claudin-2 Expression
Abstract
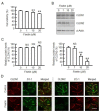
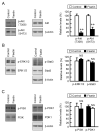
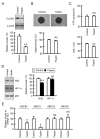
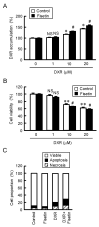
Claudin-2 (CLDN2), a component of tight junction, is involved in the reduction of anticancer drug-induced toxicity in spheroids of A549 cells derived from human lung adenocarcinoma. Fisetin, a dietary flavonoid, inhibits cancer cell growth, but its effect on chemosensitivity in spheroids is unknown. Here, we found that fisetin (20 μM) decreases the protein level of CLDN2 to 22.3%. Therefore, the expression mechanisms were investigated by real-time polymerase chain reaction and Western blotting. Spheroids were formed in round-bottom plates, and anticancer drug-induced toxicity was measured by ATP content. Fisetin decreased the phosphorylated-Akt level, and CLDN2 expression was decreased by a phosphatidylinositol 3-kinase (PI3K) inhibitor, suggesting the inhibition of PI3K/Akt signal is involved in the reduction of CLDN2 expression. Hypoxia level, one of the hallmarks of tumor microenvironment, was reduced by fisetin. Although fisetin did not change hypoxia inducible factor-1α level, it decreased the protein level of nuclear factor erythroid 2-related factor 2, a stress response factor, by 25.4% in the spheroids. The toxicity of doxorubicin (20 μM) was enhanced by fisetin from 62.8% to 40.9%, which was rescued by CLDN2 overexpression (51.7%). These results suggest that fisetin can enhance anticancer drug toxicity in A549 spheroids mediated by the reduction of CLDN2 expression.
Keywords: anticancer drug toxicity; claudin-2; fisetin; lung adenocarcinoma.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures



References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

