Heterogeneities in Ventricular Conduction Following Treatment with Heptanol: A Multi-Electrode Array Study in Langendorff-Perfused Mouse Hearts
- PMID: 35888085
- PMCID: PMC9321110
- DOI: 10.3390/life12070996
Heterogeneities in Ventricular Conduction Following Treatment with Heptanol: A Multi-Electrode Array Study in Langendorff-Perfused Mouse Hearts
Abstract
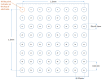
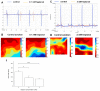
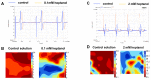
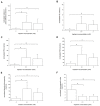
Background: Previous studies have associated slowed ventricular conduction with the arrhythmogenesis mediated by the gap junction and sodium channel inhibitor heptanol in mouse hearts. However, they did not study the propagation patterns that might contribute to the arrhythmic substrate. This study used a multi-electrode array mapping technique to further investigate different conduction abnormalities in Langendorff-perfused mouse hearts exposed to 0.1 or 2 mM heptanol. Methods: Recordings were made from the left ventricular epicardium using multi-electrode arrays in spontaneously beating hearts during right ventricular 8 Hz pacing or S1S2 pacing. Results: In spontaneously beating hearts, heptanol at 0.1 and 2 mM significantly reduced the heart rate from 314 ± 25 to 189 ± 24 and 157 ± 7 bpm, respectively (ANOVA, p < 0.05 and p < 0.001). During regular 8 Hz pacing, the mean LATs were increased by 0.1 and 2 mM heptanol from 7.1 ± 2.2 ms to 19.9 ± 5.0 ms (p < 0.05) and 18.4 ± 5.7 ms (p < 0.05). The standard deviation of the mean LATs was increased from 2.5 ± 0.8 ms to 10.3 ± 4.0 ms and 8.0 ± 2.5 ms (p < 0.05), and the median of phase differences was increased from 1.7 ± 1.1 ms to 13.9 ± 7.8 ms and 12.1 ± 5.0 ms by 0.1 and 2 mM heptanol (p < 0.05). P5 took a value of 0.2 ± 0.1 ms and was not significantly altered by heptanol at 0.1 or 2 mM (1.1 ± 0.9 ms and 0.9 ± 0.5 ms, p > 0.05). P50 was increased from 7.3 ± 2.7 ms to 24.0 ± 12.0 ms by 0.1 mM heptanol and then to 22.5 ± 7.5 ms by 2 mM heptanol (p < 0.05). P95 was increased from 1.7 ± 1.1 ms to 13.9 ± 7.8 ms by 0.1 mM heptanol and to 12.1 ± 5.0 ms by 2 mM heptanol (p < 0.05). These changes led to increases in the absolute inhomogeneity in conduction (P5−95) from 7.1 ± 2.6 ms to 31.4 ± 11.3 ms, 2 mM: 21.6 ± 7.2 ms, respectively (p < 0.05). The inhomogeneity index (P5−95/P50) was significantly reduced from 3.7 ± 1.2 to 3.1 ± 0.8 by 0.1 mM and then to 3.3 ± 0.9 by 2 mM heptanol (p < 0.05). Conclusion: Increased activation latencies, reduced CVs, and the increased inhomogeneity index of conduction were associated with both spontaneous and induced ventricular arrhythmias.
Keywords: action potential duration; conduction; detrended fluctuation analysis; dispersion; entropy; heptanol; heterogeneity; hypokalemia; inhomogeneity; mouse; variability.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Tolppanen H., Siirila-Waris K., Harjola V.P., Marono D., Parenica J., Kreutzinger P., Nieminen T., Pavlusova M., Tarvasmaki T., Twerenbold R., et al. Ventricular conduction abnormalities as predictors of long-term survival in acute de novo and decompensated chronic heart failure. ESC Heart Fail. 2016;3:35–43. doi: 10.1002/ehf2.12068. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

