The Advances of Broad-Spectrum and Hot Anti-Coronavirus Drugs
- PMID: 35889013
- PMCID: PMC9317368
- DOI: 10.3390/microorganisms10071294
The Advances of Broad-Spectrum and Hot Anti-Coronavirus Drugs
Abstract
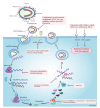
Coronaviruses, mainly including severe acute respiratory syndrome virus, severe acute respiratory syndrome coronavirus 2, Middle East respiratory syndrome virus, human coronavirus OC43, chicken infectious bronchitis virus, porcine infectious gastroenteritis virus, porcine epidemic diarrhea virus, and murine hepatitis virus, can cause severe diseases in humans and livestock. The severe acute respiratory syndrome coronavirus 2 is infecting millions of human beings with high morbidity and mortality worldwide, and the multiplicity of swine epidemic diarrhea coronavirus in swine suggests that coronaviruses seriously jeopardize the safety of public health and that therapeutic intervention is urgently needed. Currently, the most effective methods of prevention and control for coronaviruses are vaccine immunization and pharmacotherapy. However, the emergence of mutated viruses reduces the effectiveness of vaccines. In addition, vaccine developments often lag behind, making it difficult to put them into use early in the outbreak. Therefore, it is meaningful to screen safe, cheap, and broad-spectrum antiviral agents for coronaviruses. This review systematically summarizes the mechanisms and state of anti-human and porcine coronavirus drugs, in order to provide theoretical support for the development of anti-coronavirus drugs and other antivirals.
Keywords: PEDV; SARS-CoV-2; TCM; antiviral drugs; coronaviruses.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Current concepts in the development of therapeutics against human and animal coronavirus diseases by targeting NP.Comput Struct Biotechnol J. 2021;19:1072-1080. doi: 10.1016/j.csbj.2021.01.032. Epub 2021 Jan 30. Comput Struct Biotechnol J. 2021. PMID: 33552444 Free PMC article. Review.
-
Melatonin and other indoles show antiviral activities against swine coronaviruses in vitro at pharmacological concentrations.J Pineal Res. 2021 Sep;71(2):e12754. doi: 10.1111/jpi.12754. Epub 2021 Jun 24. J Pineal Res. 2021. PMID: 34139040
-
Nanoparticle-Based Strategies to Combat COVID-19.ACS Appl Nano Mater. 2020 Aug 25;3(9):8557-8580. doi: 10.1021/acsanm.0c01978. eCollection 2020 Sep 25. ACS Appl Nano Mater. 2020. PMID: 37556239 Review.
-
Coronavirus Endoribonuclease Activity in Porcine Epidemic Diarrhea Virus Suppresses Type I and Type III Interferon Responses.J Virol. 2019 Apr 3;93(8):e02000-18. doi: 10.1128/JVI.02000-18. Print 2019 Apr 15. J Virol. 2019. PMID: 30728254 Free PMC article.
-
Vaccines for porcine epidemic diarrhea virus and other swine coronaviruses.Vet Microbiol. 2017 Jul;206:45-51. doi: 10.1016/j.vetmic.2016.11.029. Epub 2016 Dec 2. Vet Microbiol. 2017. PMID: 27964998 Free PMC article. Review.
Cited by
-
Lessons learnt from broad-spectrum coronavirus antiviral drug discovery.Expert Opin Drug Discov. 2024 Sep;19(9):1023-1041. doi: 10.1080/17460441.2024.2385598. Epub 2024 Jul 30. Expert Opin Drug Discov. 2024. PMID: 39078037 Review.
-
Advances research in porcine enteric coronavirus therapies and antiviral drugs.Vet Q. 2024 Dec;44(1):1-49. doi: 10.1080/01652176.2024.2421299. Epub 2024 Nov 1. Vet Q. 2024. PMID: 39484691 Free PMC article. Review.
-
Increased viperin expression induced by avian infectious bronchitis virus inhibits viral replication by restricting cholesterol synthesis: an in vitro study.Vet Res. 2024 Sep 27;55(1):116. doi: 10.1186/s13567-024-01368-w. Vet Res. 2024. PMID: 39334500 Free PMC article.
-
Antiviral activity of chrysin and naringenin against porcine epidemic diarrhea virus infection.Front Vet Sci. 2023 Dec 7;10:1278997. doi: 10.3389/fvets.2023.1278997. eCollection 2023. Front Vet Sci. 2023. PMID: 38130439 Free PMC article.
-
Mechanism of PRRSV infection and antiviral role of polyphenols.Virulence. 2024 Dec;15(1):2417707. doi: 10.1080/21505594.2024.2417707. Epub 2024 Oct 21. Virulence. 2024. PMID: 39432383 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

