Complex Trophic Interactions in an Acidophilic Microbial Community
- PMID: 35889059
- PMCID: PMC9321944
- DOI: 10.3390/microorganisms10071340
Complex Trophic Interactions in an Acidophilic Microbial Community
Abstract
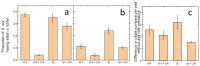
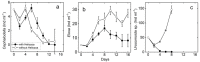
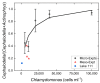
Extreme habitats often harbor specific communities that differ substantially from non-extreme habitats. In many cases, these communities are characterized by archaea, bacteria and protists, whereas the number of species of metazoa and higher plants is relatively low. In extremely acidic habitats, mostly prokaryotes and protists thrive, and only very few metazoa thrive, for example, rotifers. Since many studies have investigated the physiology and ecology of individual species, there is still a gap in research on direct, trophic interactions among extremophiles. To fill this gap, we experimentally studied the trophic interactions between a predatory protist (Actinophrys sol, Heliozoa) and its prey, the rotifers Elosa woralli and Cephalodella sp., the ciliate Urosomoida sp. and the mixotrophic protist Chlamydomonas acidophila (a green phytoflagellate, Chlorophyta). We found substantial predation pressure on all animal prey. High densities of Chlamydomonas acidophila reduced the predation impact on the rotifers by interfering with the feeding behaviour of A. sol. These trophic relations represent a natural case of intraguild predation, with Chlamydomonas acidophila being the common prey and the rotifers/ciliate and A. sol being the intraguild prey and predator, respectively. We further studied this intraguild predation along a resource gradient using Cephalodella sp. as the intraguild prey. The interactions among the three species led to an increase in relative rotifer abundance with increasing resource (Chlamydomonas) densities. By applying a series of laboratory experiments, we revealed the complexity of trophic interactions within a natural extremophilic community.
Keywords: Rotifera; acid mine drainage; extremophiles; food web; heliozoa; intraguild predation; mining lakes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Vertical niche separation of two consumers (Rotatoria) in an extreme habitat.Oecologia. 2004 May;139(4):594-603. doi: 10.1007/s00442-004-1545-z. Epub 2004 Mar 31. Oecologia. 2004. PMID: 15054658
-
Spatial refuge from intraguild predation: implications for prey suppression and trophic cascades.Oecologia. 2006 Aug;149(2):265-75. doi: 10.1007/s00442-006-0443-y. Epub 2006 May 18. Oecologia. 2006. PMID: 16708227
-
Multiple environmental stressors confine the ecological niche of the rotifer Cephalodella acidophila.Freshw Biol. 2013 May;58(5):1008-1015. doi: 10.1111/fwb.12104. Freshw Biol. 2013. PMID: 23704795 Free PMC article.
-
Habitat structure affects intraguild predation.Ecology. 2007 Nov;88(11):2713-9. doi: 10.1890/06-1408.1. Ecology. 2007. PMID: 18051638 Review.
-
Predators, parasitoids, and pathogens: a cross-cutting examination of intraguild predation theory.Ecology. 2007 Nov;88(11):2681-8. doi: 10.1890/06-1707.1. Ecology. 2007. PMID: 18051634 Review.
Cited by
-
Eukaryotic Community Structure and Interspecific Interactions in a Stratified Acidic Pit Lake Water in Anhui Province.Microorganisms. 2023 Apr 9;11(4):979. doi: 10.3390/microorganisms11040979. Microorganisms. 2023. PMID: 37110402 Free PMC article.
References
-
- Bell E.M. Life at Extremes–Environments, Organisms and Strategies for Survival. CABI; Wallingford, UK: 2012. 554p
-
- Nixdorf B., Lessmann D., Deneke R. Mining lakes in a disturbed landscape: Application of the EC Water Framework Directive and future management strategies. Ecol. Eng. 2005;24:67–73. doi: 10.1016/j.ecoleng.2004.12.008. - DOI
-
- Pedrozo F., Kelly L., Diaz M., Temporetti P., Baffico G., Kringel R., Friese K., Mages M., Geller W., Woelfl S. First results on the water chemistry, algae and trophic status of an Andean acidic lake system of volcanic origin in Patagonia (Lake Caviahue) Hydrobiologia. 2001;452:129–137. doi: 10.1023/A:1011984212798. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

