The Phytochemistry and Pharmacology of Tulbaghia, Allium, Crinum and Cyrtanthus: 'Talented' Taxa from the Amaryllidaceae
- PMID: 35889346
- PMCID: PMC9316996
- DOI: 10.3390/molecules27144475
The Phytochemistry and Pharmacology of Tulbaghia, Allium, Crinum and Cyrtanthus: 'Talented' Taxa from the Amaryllidaceae
Abstract
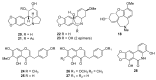
Amaryllidaceae is a significant source of bioactive phytochemicals with a strong propensity to develop new drugs. The genera Allium, Tulbaghia, Cyrtanthus and Crinum biosynthesize novel alkaloids and other phytochemicals with traditional and pharmacological uses. Amaryllidaceae biomolecules exhibit multiple pharmacological activities such as antioxidant, antimicrobial, and immunomodulatory effects. Traditionally, natural products from Amaryllidaceae are utilized to treat non-communicable and infectious human diseases. Galanthamine, a drug from this family, is clinically relevant in treating the neurocognitive disorder, Alzheimer's disease, which underscores the importance of the Amaryllidaceae alkaloids. Although Amaryllidaceae provide a plethora of biologically active compounds, there is tardiness in their development into clinically pliable medicines. Other genera, including Cyrtanthus and Tulbaghia, have received little attention as potential sources of promising drug candidates. Given the reciprocal relationship of the increasing burden of human diseases and limited availability of medicinal therapies, more rapid drug discovery and development are desirable. To expedite clinically relevant drug development, we present here evidence on bioactive compounds from the genera Allium, Tulgbaghia, Cyrtanthus and Crinum and describe their traditional and pharmacological applications.
Keywords: Allium; Amaryllidaceae; Crinum; Cyrtanthus; Tulbaghia; alkaloids; drug discovery; natural products; pharmacological activity; phytochemicals.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


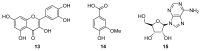
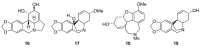
References
-
- Chase M.W., Reveal J.L., Fay M.F. A subfamilial classification for the expanded asparagalean families Amaryllidaceae, Asparagaceae and Xanthorrhoeaceae. Bot. J. Linn. Soc. 2009;161:132–136. doi: 10.1111/j.1095-8339.2009.00999.x. - DOI
-
- Vosa C.G., Siebert S.J., Van Wyk A.E.B. Micromorphology and cytology of Prototulbaghia siebertii, with notes on its taxonomic significance. Upsp. Inst. Repos. 2011;41:311–314.
-
- Elgorashi E.E., van Staden J. Bioactivity and Bioactive Compounds of African Amaryllidaceae. ACS Publications; Washington, DC, USA: 2009.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

